Advertisement
Advertisement
March/April 2024
The Continued Role of Transesophageal Echocardiography in TAVR: When a Minimalistic Approach Is Not Enough
A review of when transesophageal echocardiography may be preferred over transthoracic echocardiography and fluoroscopy during TAVR procedures.
Since the first-ever case of transcatheter aortic valve replacement (TAVR) in 2002, the technique and procedural requirements for performing a TAVR procedure have changed dramatically. This is in part due to the experience gained from the early TAVR cases as well as the advancement of valve design and improvements in delivery systems. Similarly, the role of intraprocedural imaging has changed over time, with early TAVR cases all performed under general anesthesia with transesophageal echocardiography (TEE) guidance to now, where the majority of cases are done under monitored anesthesia care (MAC) with transthoracic echocardiography (TTE) guidance.1-3 Furthermore, MAC has been shown to be associated with shorter procedural time and shorter length of stay.4 More than half (60.3%) of all TAVR cases in the United States were done under MAC from 2011 to 2018, according to the TVT registry.5 However, this has changed since that time, with a higher proportion of sites utilizing conscious sedation, with its use increasing from 50% to 76% and the proportion of patients receiving conscious sedation increasing from 33% to 64%. This is due to increasing experience and comfort level by operators at various institutions performing TAVR and studies showing improved outcomes seen with utilization of conscious sedation.6 TEE in MAC cases is now reserved as a backup in as-needed situations.
TAVR is routinely executed with the aid of multiple imaging modalities. TTE is used for the initial diagnosis of significant aortic stenosis (AS) or aortic regurgitation (AR). Multidetector CT (MDCT) is then used for further assessment and procedural planning to evaluate sizing, distribution of calcification, and access site considerations. Fluoroscopy is almost always performed at the time of TAVR intervention, frequently in conjunction with aortogram as well as peripheral and coronary angiography. Additional imaging modalities also play a significant role in selected cases. These modalities include TEE and cardiac magnetic resonance imaging (CMRI). This article explores the continued utilization and relevance of TEE in the current TAVR era.
USE OF TRANSESOPHAGEAL IMAGING WHEN TRANSTHORACIC IMAGING IS SUBOPTIMAL
Transthoracic imaging is routinely used for the majority of uncomplicated TAVR procedures. However, occasionally TTE imaging falls short in providing adequate assistance during intraprocedural imaging. TEE has greater spatial resolution when compared to TTE. It can override the common challenges faced by TTE during intraprocedural imaging, such as supine imaging instead of left lateral, barrel-shaped chest due to emphysematous lungs in chronic obstructive pulmonary disease, radiation-related AS in patients with breast implants, and TAVR in patients with chest wall deformities like pectus excavatum. Factors that might urge providers to consider upfront TEE instead of the routinely performed TTE are described in Table 1.
Furthermore, important preassessment details such as presence of AR, other coexisting valvular pathology, intracardiac thrombus or significant aortic atheroma can be overlooked with poor TTE imaging windows. One of the most important markers of procedural success is the absence of paravalvular leak (PVL). Suboptimal imaging windows may preclude assessment of PVL and may underestimate the degree of paravalvular regurgitation. More than mild (1+) PVL is directly related to poor outcomes, according to multiple prospective studies.7-9 The accurate location and degree of PVL cannot be assessed with certainty with an aortogram under fluoroscopy alone. Figure 1 shows the various examples of challenges faced with TTE imaging in certain patient populations.
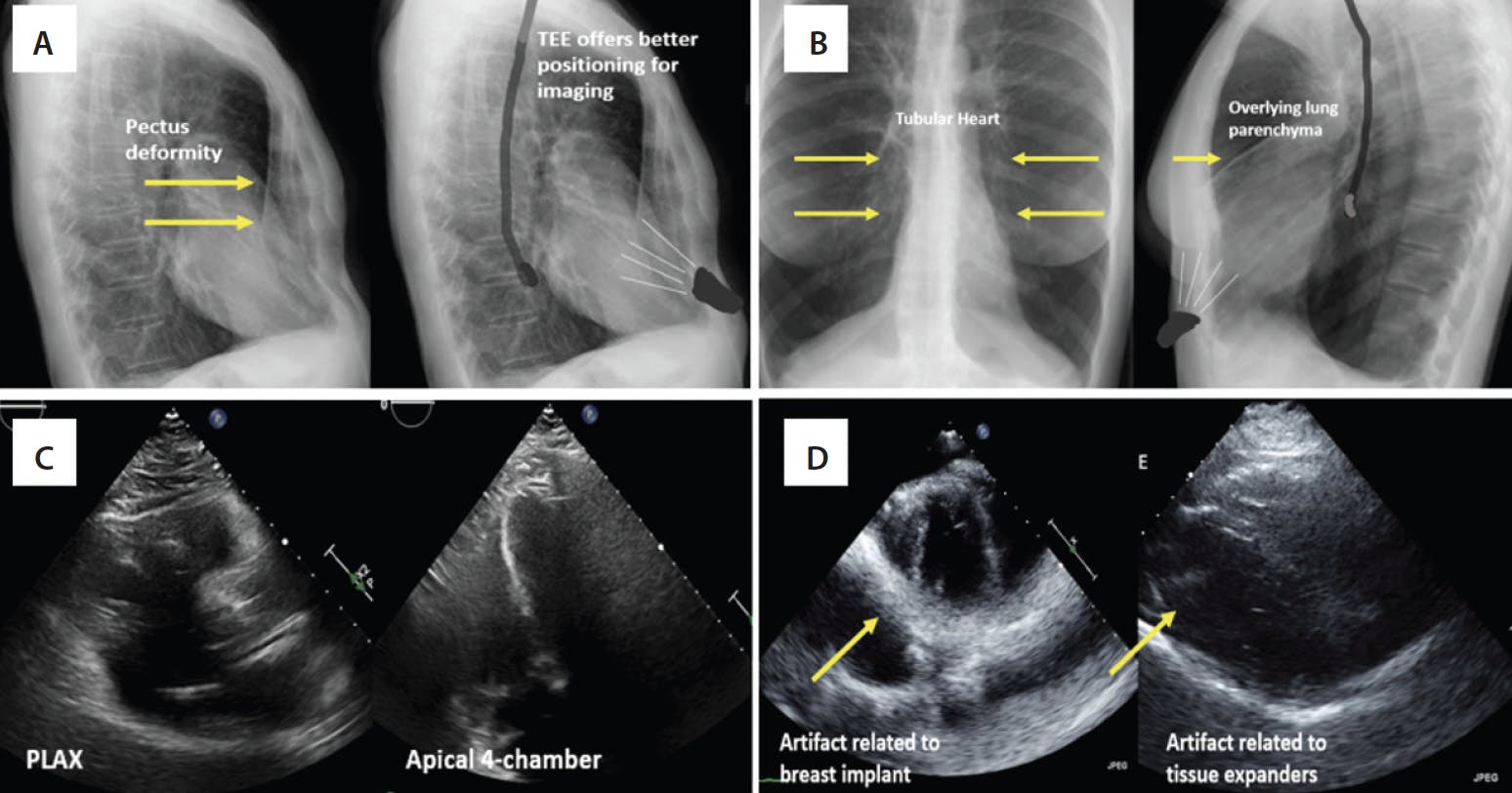
Figure 1. Chest x-ray of a patient with a pectus deformity showing the sternal distortion. The position of TTE and TEE is shown in relation to the heart (A). Chest x-ray and relative positioning of probes in a patient with chronic obstructive pulmonary disease and a tubular heart and overinflated lungs (B). Suboptimal parasternal long axis (PLAX) and apical four-chamber imaging windows in a patient undergoing TAVR (C). Patient with breast implants showing significant artifact preventing visualization of aortic valve (D).
High-Risk Native Valve Anatomy
Severe bulky asymmetrical calcification of the leaflets, sinotubular junction, or left ventricular outflow tract (LVOT) and annulus poses a significant risk for complications associated with the TAVR procedure. The complications include aortic root injury or annular rupture, moderate or more PVL, incomplete expansion of the TAVR valve, structural valve degeneration (SVD), need for permanent pacemaker, and stroke. The risk for these complications is higher in patients with bicuspid aortic valve (BAV) than tricuspid aortic valve morphology.10,11 Historically, BAV patients were excluded from pivotal trials due to complex and variable anatomy, coexisting aortopathy, and young age of BAV patients. However, the practice of offering TAVR as an option for BAV patients has increased over time. If there is reasonable anatomy, TAVR is a safe and efficacious option for BAV patients based on results from recent studies.12,13 As we move into an era in which TAVR is considered for the low-risk AS population, caution should be exercised with careful patient selection, preprocedural planning, and close intraprocedural monitoring to avoid the aforementioned complications.
Optimal imaging is key in preventing complications during high-risk TAVR procedures. Simultaneous imaging during ongoing fluoroscopy is only possible with TEE at the time of TAVR. “Poking” or protrusion of calcium during valve deployment can be visualized, and the interventionalist is alerted of this finding. For sinotubular calcification, the goal may be to land just below the hunk of calcification, which is also easily done with TEE guidance. The decision to postdilate the implanted TAVR valve in cases of moderate or more PVL and underexpanded valves is also made instantaneously, and this can be performed in a safe manner with continuous imaging. Inability to visualize acute complications may lead to delay in diagnosis and intervention leading to grim consequences. Figure 2 demonstrates the various high-risk features, TAVR complications, and use of TEE during intraprocedural imaging for high-risk TAVR anatomy.

Figure 2. Spiculated LVOT calcification seen on preassessment (yellow arrows) (A). Intraprocedural TEE demonstrating protrusion of calcium into the surrounding tissue (red arrows) (B). Orthogonal imaging of the aorta showing a dissection flap seen in a patient with dissection following a TAVR (white arrow) (C). Example of moderate PVL (green arrow) related to under-expansion of TAVR valve due to calcification that was treated with postdilatation (D, E). Erosion of large descending aortic atheroma noted on TEE (orange arrows) (F).
Coronary Artery Obstruction During TAVR
Coronary obstruction during TAVR is a devastating complication that occurs when the newly implanted valve displaces the native or prior degenerated prosthetic valve leaflet over the coronary ostium, leading to obstruction of blood flow into the vessel. Coronary obstruction is associated with grave consequences, with mortality exceeding 50%.14
The BASILICA (bioprosthetic or native aortic scallop intentional laceration to prevent coronary artery obstruction) technique was developed and presented in 2018 as an alternative to the previous bailout strategies in case of acute coronary occlusion, which included emergency bypass surgery or attempt to percutaneously intervene.15 Prior to the routine use of MDCT for preprocedural planning, this complication was often detected after the TAVR valve was implanted, subsequently leading to delays in intervention. Currently, patients at high risk for coronary obstruction are identified during preprocedural CT evaluation so a planned intervention can be performed at the time of TAVR.16,17 Coronary obstruction is much more common in valve-in-valve (ViV) TAVR. This is often due to reduction in sinus space after surgical bioprosthetic valve implantation and supra-annular implantation of the surgical valve. Some bioprosthetic valves have leaflets sewn outside the stent frame to allow for maximal effective orifice area (eg, Crown [Sorin Group], Dokimos [Labcor], Mitroflow [Sorin Group], Trifecta and Trifecta GT valve [both Abbott]). These valves carry a higher risk for coronary obstruction during a ViV procedure.
With the help of multiplanar imaging and three-dimensional (3D) imaging at the time of TAVR, TEE helps identify the left main occlusion at the time of balloon inflation for borderline cases, guide snorkel procedures, and identify the laceration point in front of the coronary ostium to guide optimal laceration of the leaflet, as well as aid use of newer investigational devices such as ShortCut (Pi-Cardia), a dedicated leaflet splitting device.18 Additionally, TEE also helps confirm the measurements obtained from CT prior to commencement of the procedure. Figure 3 shows examples of the use of TEE in coronary protection.
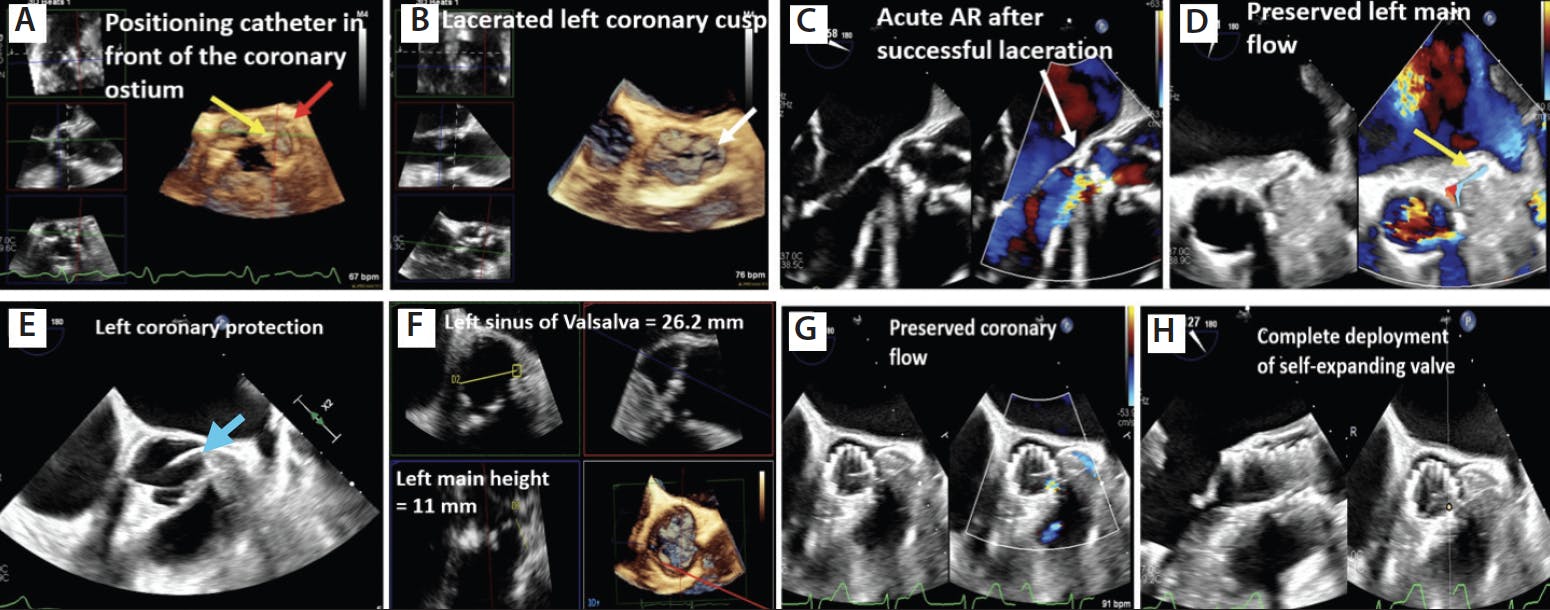
Figure 3. Three-dimensional multiplanar imaging showing position of the BASILICA catheter (yellow arrow) in front of the left main coronary artery (red arrow) (A). Laceration of the left coronary cusp during BASILICA procedure (white arrow) (B). Color-compare imaging of the aortic valve showing acute AR following leaflet laceration (white arrow) (C). Preserved coronary blood flow noted in the left coronary system following valve implantation (D). Example of coronary protection with prewiring of the coronary artery (blue arrow) (E). Measurement of the sinus dimensions using multiplanar reconstruction (MPR) imaging in a case with borderline sinus measurement (F). TEE short-axis image demonstrating preserved space in the sinus and flow into the left coronary (G). The self-expanding valve was completely deployed under constant visualization (H).
Patient characteristics at highest risk for coronary obstruction, as demonstrated in multiple prior studies, are female sex, coronary height < 10 mm, sinus of Valsalva < 28 mm, bulky calcification, long leaflet extending beyond the coronary ostia, valve-to-coronary ostium distance ≤ 4 mm for ViV TAVR, and prosthetic valves with leaflets sewn on the outside of the stent frame.
Alternative Access Requiring Surgical Preparation of the Chest
TAVR via transfemoral access is considered the gold standard approach, but this approach is not feasible in approximately 5% to 10% of patients due to peripheral vascular challenges such as severe iliofemoral calcification, suboptimal vascular size, certain peripheral interventions, or severe vascular tortuosity that prohibits the passage of catheters.1,2 To avoid the use of transfemoral access in these patients, alternative access techniques have been developed, including transcaval, transcarotid, subclavian, direct aortic/suprasternal, and transaxillary access. Most alternative access routes need surgical preparation of the patient’s chest and neck, which precludes transthoracic imaging at the time of valve intervention with the imager positioned on the left side of the patient. Imaging with TEE offers an ergonomic solution to this problem. TEE can help guide alternative access TAVR with the imager stationed at the head or right of the patient. Figure 4 shows the various access points used for TAVR and the arrangement of personnel in the operating room for alternative access cases.
ViV for Poor Fluoroscopically Visualized Valves
ViV TAVR is an attractive alternative to redo surgery for treating patients with failed prior aortic bioprosthesis who are at high risk for surgery.19 Appropriate selection of valve type, size, intraprocedural placement, and adequate expansion are all key elements for successful ViV implantation. Favorable outcomes (ie, lower rate of moderate to severe PVL, lower transvalvular gradients, lower PVL incidence) depend on all the previously mentioned factors.20,21 Most surgical bioprosthetic valve have their stent frame as a fluoroscopic marker, which allows for placement using fluoroscopy. However, there are certain valves with poor or no fluoroscopic markers, which makes landing of the TAVR valve challenging with fluoroscopy alone (Figure 5). In these cases, TEE is extremely helpful in simultaneous visualization, with two-dimensional and 3D imaging used to land the TAVR valve at the desired location and ensuring complete expansion (Figure 6).

Figure 6. Color Doppler image of the aortic valve showing degenerated bioprosthesis with severe eccentric AR (yellow arrow) (A). Three-dimensional MPR image showing positioning of the TAVR for ViV deployment (B). Fluoroscopic image of TAVR within a degenerated Biocor Epic valve (Abbott). Biocor valve has poor visualization on fluoroscopy (C). Positioning of TAVR valve in a patient with a bioroot (D). Final position showing good TAVR placement (E). Fluoroscopic image showing lack of markers during TAVR valve deployment (F).
TAVR With Minimal or No Contrast
Early studies from the various databases revealed an incidence of chronic kidney disease (CKD) of 33.5% and end-stage renal disease of 4.1% in patients undergoing TAVR.22,23 Presence of renal disease modifies the natural progression of various cardiovascular diseases due to accelerated calcium deposition and early degeneration of valves. Advanced renal disease has been shown to have poor short- and long-term outcomes after TAVR.22,24 Iodinated contrast is used in several stages of planning and executing TAVR such as the preprocedural CT scan, access at the time of TAVR, positioning of the TAVR valve, aortography for assessment of PVL, and assessment of peripheral vasculature post-TAVR.
All these steps increase the likelihood of acute kidney injury at the time of the procedure. Experienced institutions have developed ways to mitigate this issue by performing TAVR with low or no contrast. The preassessment needed for sizing of the TAVR valve can be done using 3D TEE, either at the time of TAVR or as a study done prior to TAVR. The use of 3D multiplane reconstruction (MPR) imaging has been validated against MDCT for assessment of aortic annular sizing using multiple available software. New automated software is being developed and some are currently in use for accurate modeling and assessment of annular and root measurements.25-28 The access in patients with advanced renal disease can be carefully performed using ultrasound guidance. Intravascular ultrasound is another tool that can be used for vessels not accessible with surface ultrasound. TEE guidance is used for valve positioning and postassessment after TAVR. Figure 7 shows an example of a patient with amyloidosis and advanced renal disease with severe symptomatic AS who underwent a TAVR using TEE and peripheral ultrasound.

Figure 7. Short-axis image of the left ventricle showing diffusely thick walls in a patient with stage 4 CKD (A). Strain imaging showing the classic “cherry on top” apical sparing appearance suggesting amyloidosis in a CKD patient (B). Peripheral vascular ultrasound used to access the common femoral artery (C). Use of the mitral valve navigator software for assessing aortic annular measurement (D). Use of 3D MPR imaging measurement of both left and right coronary height and leaflet length (E). Use of orthogonal imaging for valve positioning prior to deployment (F). 2D, two-dimensional; LV, left ventricle; MVN, mitral valve navigator.
Evaluation of New Devices for Treatment of AS and AR
There is constant evolution and innovation in the field of structural heart disease. Multiple new TAVR devices are being developed, and there are ongoing efforts in the industry to make a durable TAVR device with lower incidence of SVD. Early iterations of these new devices may need the aid of TEE at the time of TAVR for assessing feasibility of valve deployment, accurate assessment of PVL, and transaortic gradients.
CONCLUSION
Although TTE in conjunction with fluoroscopy is the predominant imaging modality used in TAVR cases performed across most institutions, there is a role for TEE in select complicated cases. Careful planning and the use of TEE proactively in these cases can avert complications.
1. Carroll JD, Mack MJ, Vemulapalli S, et al. STS-ACC TVT registry of transcatheter aortic valve replacement. Ann Thorac Surg. 2021;111:701-722. doi: 10.1016/j.athoracsur.2020.09.002
2. Carroll JD, Mack MJ, Vemulapalli S, et al. STS-ACC TVT registry of transcatheter aortic valve replacement. J Am Coll Cardiol. 2020;76:2492-2516. doi: 10.1016/j.jacc.2020.09.595
3. Mack MJ, Brennan JM, Brindis R, et al. Outcomes following transcatheter aortic valve replacement in the United States. JAMA. 2013;310:2069-2077. doi: 10.1001/jama.2013.282043
4. Eskandari M, Aldalati O, Dworakowski R, et al. Comparison of general anaesthesia and non-general anaesthesia approach in transfemoral transcatheter aortic valve implantation. Heart. 2018;104:1621-1628. doi: 10.1136/heartjnl-2017-312559
5. Arnold SV, Manandhar P, Vemulapalli S, et al. Mediators of improvement in TAVR outcomes over time: insights from the STS-ACC TVT registry. Circ Cardiovasc Interv. 2023;16:e013080. doi: 10.1161/CIRCINTERVENTIONS.123.013080
6. Butala NM, Chung M, Secemsky EA, et al. Conscious sedation versus general anesthesia for transcatheter aortic valve replacement: variation in practice and outcomes. JACC Cardiovasc Interv. 2020;13:1277-1287. doi: 10.1016/j.jcin.2020.03.008
7. Okuno T, Tomii D, Heg D, et al. Five-year outcomes of mild paravalvular regurgitation after transcatheter aortic valve implantation. EuroIntervention. 2022;18:33-42. doi: 10.4244/EIJ-D-21-00784
8. Laakso T, Laine M, Moriyama N, et al. Impact of paravalvular regurgitation on the mid-term outcome after transcatheter and surgical aortic valve replacement. Eur J Cardiothorac Surg. 2020;58:1145-1152. doi: 10.1093/ejcts/ezaa254
9. Pibarot P, Hahn RT, Weissman NJ, et al. Association of paravalvular regurgitation with 1-year outcomes after transcatheter aortic valve replacement with the SAPIEN 3 valve. JAMA Cardiol. 2017;2:1208-1216. doi: 10.1001/jamacardio.2017.3425
10. Gupta R, Mahmoudi E, Behnoush AH, et al. Clinical outcomes and the impact of valve morphology for transcatheter aortic valve replacement in bicuspid aortic valves: a systematic review and meta-analysis. Catheter Cardiovasc Interv. 2023;102:721-730. doi: 10.1002/ccd.30808
11. Nuyens P, DeBacker O, Sathananthan J, et al. TAVR in bicuspid aortic stenosis: current evidence and proposal for a randomized controlled trial design. JACC Cardiovasc Interv. 2023;16:1682-1687. doi: 10.1016/j.jcin.2023.04.003
12. Deeb GM, Reardon MJ, Ramlawi B, et al. Propensity-matched 1-year outcomes following transcatheter aortic valve replacement in low-risk bicuspid and tricuspid patients. JACC Cardiovasc Interv. 2022;15:511-522. doi: 10.1016/j.jcin.2021.10.027
13. Fiorina C, Massussi M, Ancona M, et al. Mid-term outcomes and hemodynamic performance of transcatheter aortic valve implantation in bicuspid aortic valve stenosis: Insights from the bicuSpid TAvi duraBILITY (STABILITY) registry. Catheter Cardiovasc Interv. 2023;102:1132-1139. doi: 10.1002/ccd.30813
14. Ribeiro HB, Rodes-Cabau J, Blanke P, et al. Incidence, predictors, and clinical outcomes of coronary obstruction following transcatheter aortic valve replacement for degenerative bioprosthetic surgical valves: insights from the VIVID registry. Eur Heart J. 2018;39:687-695. doi: 10.1093/eurheartj/ehx455
15. Khan JM, Dvir D, Greenbaum AB, et al. Transcatheter laceration of aortic leaflets to prevent coronary obstruction during transcatheter aortic valve replacement: concept to first-in-human. JACC Cardiovasc Interv. 2018;11:677-689. doi: 10.1016/j.jcin.2018.01.247
16. Dvir D, Leipsic J, Blanke P, et al. Coronary obstruction in transcatheter aortic valve-in-valve implantation: preprocedural evaluation, device selection, protection, and treatment. Circ Cardiovasc Interv. 2015;8:e002079. doi: 10.1161/CIRCINTERVENTIONS.114.002079
17. Ribeiro HB, Webb JG, Makkar RR, et al. Predictive factors, management, and clinical outcomes of coronary obstruction following transcatheter aortic valve implantation: insights from a large multicenter registry. J Am Coll Cardiol. 2013;62:1552-1562. doi: 10.1016/j.jacc.2013.07.040
18. Dvir D, Leon MB, Abdel-Wahab M, et al. First-in-human dedicated leaflet splitting device for prevention of coronary obstruction in transcatheter aortic valve replacement. JACC Cardiovasc Interv. 2023;16:94-102. doi: 10.1016/j.jcin.2022.10.050
19. Tarantini G, Sathananthan J, Fabris T, et al. Transcatheter aortic valve replacement in failed transcatheter bioprosthetic valves. JACC Cardiovasc Interv. 2022;15:1777-1793. doi: 10.1016/j.jcin.2022.07.035
20. Alperi A, Garcia S, Rodes-Cabau J. Transcatheter valve-in-valve implantation in degenerated surgical aortic and mitral bioprosthesis: current state and future perspectives. Prog Cardiovasc Dis. 2022;72:54-65. doi: 10.1016/j.pcad.2021.10.001
21. Nuche J, Abbas AE, Serra V, et al. Balloon- vs self-expanding transcatheter valves for failed small surgical aortic bioprostheses: 1-year results of the LYTEN trial. JACC Cardiovasc Interv. 2023;16:2999-3012. doi: 10.1016/j.jcin.2023.10.028
22. Gupta T, Goel K, Kolte D, et al. Association of chronic kidney disease with in-hospital outcomes of transcatheter aortic valve replacement. JACC Cardiovasc Interv. 2017;10:2050-2060. doi: 10.1016/j.jcin.2017.07.044
23. Allende R, Webb JG, Munoz-Garcia AJ, et al. Advanced chronic kidney disease in patients undergoing transcatheter aortic valve implantation: insights on clinical outcomes and prognostic markers from a large cohort of patients. Eur Heart J. 2014;35:2685-2696. doi: 10.1093/eurheartj/ehu175
24. Naser JA, Luis SA, Pislaru SV, et al. Impact on kidney function and medium-term outcomes of transcatheter aortic valve replacement in patients with chronic kidney disease. Am J Cardiol. 2024;210:163-171. doi: 10.1016/j.amjcard.2023.10.014
25. Hana D, Miller T, Skaff P, et al. 3D transesophageal echocardiography for guiding transcatheter aortic valve replacement without prior cardiac computed tomography in patients with renal dysfunction. Cardiovasc Revasc Med. 2022;41:63-68. doi: 10.1016/j.carrev.2021.12.026
26. Khalique OK, Kodali SK, Paradis JM, et al. Aortic annular sizing using a novel 3-dimensional echocardiographic method: use and comparison with cardiac computed tomography. Circ Cardiovasc Imaging. 2014;7:155-163. doi: 10.1161/CIRCIMAGING.113.001153
27. Prihadi EA, van Rosendael PJ, Vollema EM, et al. Feasibility, accuracy, and reproducibility of aortic annular and root sizing for transcatheter aortic valve replacement using novel automated three-dimensional echocardiographic software: comparison with multi-detector row computed tomography. J Am Soc Echocardiogr. 2018;31:505-514.e3. doi: 10.1016/j.echo.2017.10.003
28. Rong LQ, Hameed I, Salemi A, et al. Three-dimensional echocardiography for transcatheter aortic valve replacement sizing: a systematic review and meta-analysis. J Am Heart Assoc. 2019;8:e013463. doi: 10.1161/JAHA.119.013463
Advertisement
Advertisement