Advertisement
Advertisement
March/April 2022
Percutaneous Treatment of Coronary Artery Disease in Patients Undergoing TAVR
Before, after, or medical therapy?
Approximately half of patients who undergo transcatheter aortic valve replacement (TAVR) have coronary artery disease (CAD).1,2 Whether and when patients need to undergo revascularization remains a subject of ongoing debate. There have been conflicting data on the impact of CAD on clinical outcomes after TAVR. Additionally, the use of varying definitions of CAD and the infrequent reporting of anatomic features and physiologic assessment of lesion severity have hampered our ability to select which patients may benefit from revascularization. In this article, we discuss the prevalence of CAD in patients undergoing evaluation for TAVR, current practices in diagnosis and management of CAD in this population, data on outcomes of revascularization in TAVR patients, and considerations for management of coronary events after TAVR.
EPIDEMIOLOGY OF CAD IN PATIENTS WITH SEVERE SYMPTOMATIC AORTIC STENOSIS
The prevalence of both CAD and aortic stenosis (AS) increases with advancing age. As compared to the cohorts of patients with prohibitive, high, and even intermediate surgical risk enrolled in early trials of TAVR, the prevalence of CAD was significantly lower in the PARTNER 3 and Evolut Low Risk trials of younger patients with low surgical risk (Figure 1).3
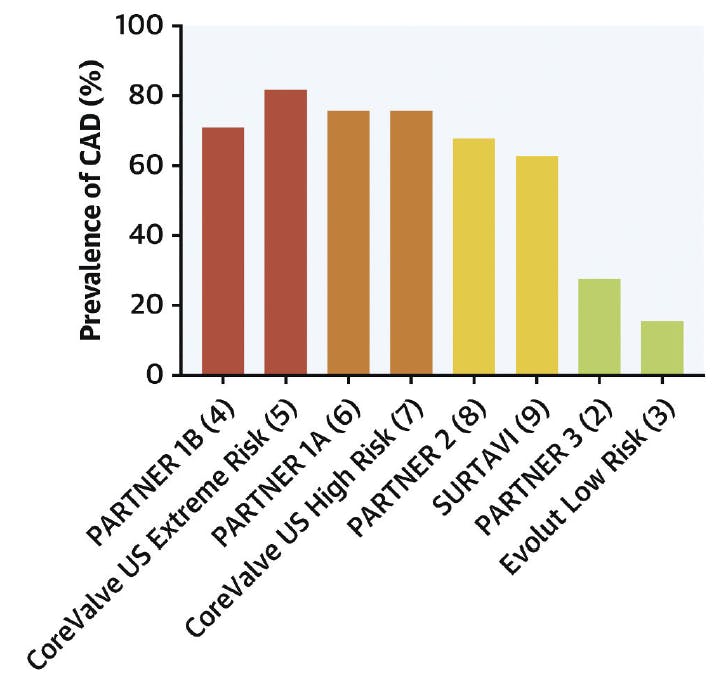
Figure 1. The prevalence of CAD in pivotal trials of TAVR. Reprinted from Faroux L, Guimaraes L, Wintzer-Wehekind J, et al. Coronary artery disease and transcatheter aortic valve replacement: JACC state-of-the-art review. J Am Coll Cardiol. 2019;74:362-372. doi: 10.1016/j.jacc.2019.06.012, with permission from Elsevier.
Historically, there has been a preference to treat significant coronary lesions with percutaneous coronary intervention (PCI), usually before or during TAVR, under the rationale that CAD could be contributing to the patient’s clinical presentation. There has also been concern over the theoretical risk of inducing ischemia and hemodynamic deterioration during TAVR in patients with severe underlying CAD.3 However, the impact of the presence of concomitant CAD in patients undergoing TAVR remains unclear, and the lack of a strong evidence base has led to considerable heterogeneity in the management of these patients.
In a meta-analysis of 15 studies and > 8,000 patients undergoing TAVR (median age of 81.3 years, 46.6% male, 48.7% with CAD), there was no significant difference in all-cause mortality between patients with and without CAD at 30 days after TAVR. However, there was a significant increase in all-cause mortality at 1 year in patients with CAD, with a cumulative odds ratio of 1.21 (95% CI, 1.07-1.36; P = .002).4
Conflicting results were reported in a subsequent meta-analysis, which found no association between the presence of CAD and 30-day and 1-year mortality after TAVR. However, patients with complex CAD as defined by a SYNTAX score > 22 had greater mortality at 1 year.5 These data suggest that there is a subset of AS patients with CAD who are at higher risk of adverse outcomes, but it remains unclear whether revascularization can meaningfully lower this risk.
DIAGNOSIS AND MANAGEMENT OF CAD IN PATIENTS UNDERGOING EVALUATION FOR TAVR
Although coronary angiography remains the standard modality for assessing the presence and severity of CAD in patients undergoing evaluation for TAVR, studies have examined the efficacy of noninvasive imaging techniques such as coronary CTA (CCTA). Chieffo et al showed that CCTA at the time of cardiac CT could be safely used as a screening tool for significant CAD, with no difference in major adverse cardiovascular and cerebrovascular events (MACCE) at 30 days and 1 year between those who only underwent CCTA versus those who subsequently had invasive angiography.6
Patients with AS were excluded from trials validating invasive physiologic indices such as fractional flow reserve (FFR) and instantaneous wave-free ratio, but data mostly from retrospective observational studies show that their use in this population is both safe and effective. Lunardi et al performed a retrospective analysis of an Italian registry of patients undergoing TAVR with concomitant CAD to determine whether physiology- versus angiography-guided revascularization was associated with better clinical outcomes. After 2 years of follow-up, patients who underwent FFR-guided revascularization had better MACCE-free survival than those who underwent PCI guided by angiography alone. The majority of intermediate coronary lesions that were incidentally found during the evaluation for TAVR were negative by FFR, and deferred intervention was not associated with increased ischemic complications either during the TAVR procedure or in subsequent follow-up.7
OUTCOMES OF REVASCULARIZATION IN PATIENTS UNDERGOING TAVR
In addition to the question of whether revascularization confers benefits over medical therapy for patients with CAD and symptomatic severe AS, there is ongoing uncertainty over the optimal timing of PCI in relation to TAVR. In a single-center retrospective analysis, 258 patients undergoing TAVR and planned PCI were further subdivided into those who underwent PCI before TAVR (n = 143), those who underwent concomitant PCI and TAVR (n = 77), and those who underwent PCI after TAVR (n = 38). There were no significant differences in procedural or 30-day outcomes among the groups, including MACCE, major bleeding, major vascular complication, and acute kidney injury. Similarly, there was no significant difference in MACCE or all-cause mortality among the groups at 2-year follow-up.8
A multicenter retrospective analysis of patients compared those who underwent PCI within the year before TAVR with those who underwent either concomitant PCI at the time of TAVR or up to 60 days afterward. In the propensity-matched analysis, there was no significant difference in MACCE rate, all-cause mortality, or repeat PCI in the latter group compared to the group who had undergone PCI prior to TAVR.9
In the only prospective randomized trial to study the impact of revascularization in patients undergoing TAVR, 235 patients in the ACTIVATION trial with severe symptomatic AS and significant CAD with Canadian Cardiovascular Society class I or II angina were assigned to receive PCI or no PCI prior to TAVR. At 1 year, rates of all-cause mortality or rehospitalization were similar between the groups, occurring in 41.5% of patients who underwent PCI and 44% of those who did not.10
Three ongoing prospective studies will further our understanding of whether select patients with significant coronary ischemia and severe AS may benefit from PCI in addition to valve replacement. The NOTION-3 randomized trial will address whether the addition of FFR-guided complete revascularization to TAVR improves clinical outcomes compared to TAVR alone. The FAITAVI randomized trial will compare outcomes of FFR- versus angiographically guided revascularization in patients with severe AS undergoing TAVR. Lastly, the COMPLETE TAVR trial will randomize 4,000 patients with significant CAD after successful TAVR to PCI versus medical therapy alone.
MANAGEMENT OF CORONARY EVENTS AFTER TAVR
Despite the lack of a standardized approach to coronary revascularization in patients undergoing TAVR, unplanned PCI after the index valve procedure is rare. In a large international registry of more than 15,000 patients, only 133 (0.9%) had an unplanned PCI after TAVR at 6-year follow-up. The incidence of PCI was highest during the first several days after TAVR, and the most common indication was acute coronary syndrome (ACS). Successful PCI was reported in 96.6% of patients, with no significant difference between those treated with balloon- versus self-expandable transcatheter heart valves (THVs) (100% vs 94.9%; P = .15).11
Similarly, Kim et al reported data from 449 patients with prior TAVR requiring urgent or emergent coronary angiography, the vast majority for ACS. The data showed that PCI was successful in 91.4% of cases, regardless of THV type. Investigators found higher rates of selective cannulation of the right coronary artery, but not the left coronary artery, in patients with shorter THV frames.12
However, multiple other studies have reported greater difficulty with coronary access in patients with supra-annular as compared to balloon-expandable THVs.13,14 Due to the taller frames and higher leaflet positioning in supra-annular THVs, the placement of a commissural post in front of or near a coronary ostium, particularly when combined with a small aortic root, can make coronary access challenging, if not impossible. Tarantini et al conducted a prospective single-center study in which coronary angiography was routinely performed after TAVR to determine whether intentional commissural alignment during placement of a supra-annular THV impacted the likelihood of successful coronary access. They found increased rates of selective coronary cannulation when Evolut (Medtronic) and Acurate Neo valves (Boston Scientific Corporation) were implanted using a commissural alignment technique, but coronary access remained more challenging with an aligned supra-annular THV than with Sapien 3 valves (Edwards Lifesciences).15
CONCLUSIONS
The optimal diagnosis and management of CAD in patients with symptomatic severe AS remain areas of active investigation. As TAVR increasingly becomes the treatment of choice for younger and lower-risk patients, there is an ongoing need for further research on which subsets of patients with AS and significant CAD may benefit from revascularization.
1. Rapp AH, Hillis LD, Lange RA, Cigarroa JE. Prevalence of coronary artery disease in patients with aortic stenosis with and without angina pectoris. Am J Cardiol. 2001;87:1216-1217;A7. doi: 10.1016/s0002-9149(01)01501-6
2. Walther T, Hamm CW, Schuler G, et al. Perioperative results and complications in 15,964 transcatheter aortic valve replacements: prospective data from the GARY Registry. J Am Coll Cardiol. 2015;65:2173-2180. doi: 10.1016/j.jacc.2015.03.034
3. Faroux L, Guimaraes L, Wintzer-Wehekind J, et al. Coronary artery disease and transcatheter aortic valve replacement: JACC state-of-the-art review. J Am Coll Cardiol. 2019;74:362-372. doi: 10.1016/j.jacc.2019.06.012
4. Sankaramangalam K, Banerjee K, Kandregula K, et al. Impact of coronary artery disease on 30-day and 1-year mortality in patients undergoing transcatheter aortic valve replacement: a meta-analysis. J Am Heart Assoc. 2017;6:e006092. doi: 10.1161/JAHA.117.006092
5. D’Ascenzo F, Verardi R, Visconti M, et al. Independent impact of extent of coronary artery disease and percutaneous revascularisation on 30-day and one-year mortality after TAVI: a meta-analysis of adjusted observational results. EuroIntervention. 2018;14:e1169-e1177. doi: 10.4244/EIJ-D-18-00098
6. Chieffo A, Giustino G, Spagnolo P, et al. Routine screening of coronary artery disease with computed tomographic coronary angiography in place of invasive coronary angiography in patients undergoing transcatheter aortic valve replacement. Circ Cardiovasc Interv. 2015;8:e002025. doi: 10.1161/CIRCINTERVENTIONS.114.002025
7. Lunardi M, Scarsini R, Venturi G, et al. Physiological versus angiographic guidance for myocardial revascularization in patients undergoing transcatheter aortic valve implantation. J Am Heart Assoc. 2019;8:e012618. doi: 10.1161/JAHA.119.012618
8. Ochiai T, Yoon SH, Flint N, et al. Timing and outcomes of percutaneous coronary intervention in patients who underwent transcatheter aortic valve implantation. Am J Cardiol. 2020;125:1361-1368. doi: 10.1016/j.amjcard.2020.01.043
9. Kumar A, Sammour Y, Reginauld S, et al. Adverse clinical outcomes in patients undergoing both PCI and TAVR: analysis from a pooled multi-center registry. Catheter Cardiovasc Interv. 2021;97:529-539. doi: 10.1002/ccd.29233
10. Patterson T, Clayton T, Dodd M, et al. ACTIVATION (percutaneous coronary intervention prior to transcatheter aortic valve implantation): a randomized clinical trial. JACC Cardiovasc Interv. 2021;14:1965-1974. doi: 10.1016/j.jcin.2021.06.041
11. Stefanini GG, Cerrato E, Pivato CA, et al. Unplanned percutaneous coronary revascularization after TAVR: a multicenter international registry. JACC Cardiovasc Interv. 2021;14:198-207. doi: 10.1016/j.jcin.2020.10.031
12. Kim WK, Pellegrini C, Ludwig S, et al. Feasibility of coronary access in patients with acute coronary syndrome and previous TAVR. JACC Cardiovasc Interv. 2021;14:1578-1590. doi: 10.1016/j.jcin.2021.05.007
13. Barbanti M, Costa G, Picci A, et al. Coronary cannulation after transcatheter aortic valve replacement: the RE-ACCESS study. JACC Cardiovasc Interv. 2020;13:2542-2555. doi: 10.1016/j.jcin.2020.07.006
14. Nai Fovino L, Scotti A, Massussi M, et al. Incidence and feasibility of coronary access after transcatheter aortic valve replacement. Catheter Cardiovasc Interv. 2020;96:E535-E541. doi: 10.1002/ccd.28720
15. Tarantini G, Nai Fovino L, Scotti A, et al. Coronary access after transcatheter aortic valve replacement with commissural alignment: the ALIGN-ACCESS study. Circ Cardiovasc Interv. 2022;15:e011045. doi: 10.1161/CIRCINTERVENTIONS.121.011045
Advertisement
Advertisement
