Advertisement
Advertisement
July/August 2021
Functional Mitral Regurgitation: Learnings and Limits of the COAPT and MITRA-FR Trials
Possible explanations for the different results between the MITRA-FR and COAPT trials.
By Daniel Grinberg, MD, PhD; Xavier Armoiry, PharmD, PhD; and Jean-François Obadia, MD, PhD
Although the association between secondary mitral regurgitation (MR) and outcome is undisputed, the beneficial impact of its correction has been debated for many years.1,2 During the last few decades, surgeons have failed to convince cardiologists that it was necessary to correct secondary MR, in particular after the disappointment of the first two randomized surgical trials led by the Cardiothoracic Surgical Trials Network.3,4 Within this scope, the publication of the MITRA-FR trial in 2018 was the logical continuity of the previous studies because it confirmed the absence of benefit of secondary MR correction.5,6 This context highlights the importance of the COAPT trial showing for the first time a decrease of all-cause mortality and heart failure hospitalizations at 24 months in the percutaneous repair group, generating a lot of controversies.7 This article discusses several explanations that have been proposed to explain the different results between these two trials.
Understanding DIFFERENCES IN TRIAL RESULTS
Technical Expertise
Both of the studies have shown that there is a learning curve. The roll-in procedures before first patient inclusion were low in both the COAPT and the MITRA-FR trials (three and five, respectively), with a mean number of enrolled patients per center and per year of 2.6 and 1.7 respectively, more often with two clips implanted per patient in COAPT, and more often with three clips implanted per patient in MITRA-FR. Technical procedural success was similar at 98% for COAPT and 95% for MITRA-FR. The reduced complication rate in COAPT was mainly due to different definitions. Therefore, differences in procedural expertise between the MITRA-FR and COAPT groups of investigators were modest and are unlikely to explain divergent results of such magnitude (Figure 1).8,9

Figure 1. Compared to other publications, the MITRA-FR and COAPT results seem to be less exposed to the “Achilles’ heel” of MR recurrence, which is rapidly growing over time after surgical mitral valve repair (mainly, downsizing annuloplasty). CTSN, Cardiothoracic Surgical Trials Network; MVA, mitral valve area. Adapted with permission from Magne J, Sénéchal M, Dumesnil JG, et al. Ischemic mitral regurgitation: a complex multifaceted disease. Cardiology. 2009;112:244-59.
Percutaneous Repair Efficacy
The procedural success was high in the two studies. The 5% MR recurrence in COAPT at 1 year was the lowest ever published for functional MR (Figure 2), but also, the 17% recurrent MR in MITRA-FR was very low when compared to other publications, possibly due to the technique itself.4,10,11 MR recurrence, known as the “Achilles’ heel” of surgical repair, is usually higher, suggesting that the edge-to-edge technique may be better suited to treat functional MR than surgical downsizing annuloplasty.

Figure 2. The death rate in the control group of COAPT is higher than in the two groups of MITRA-FR, suggesting that we cannot explain the different results of the two studies by the fact that MITRA-FR patients were too severally ill due to too advanced diseases, had too severe LV dysfunction, and that it was too late to correct the MR.
Courtesy of Dr. Martin Connock (University of Warwick)
Regurgitant Surface
Differences between European Society of Cardiology and American College of Cardiology/American Heart Association definitions of secondary MR led to different inclusion criteria, resulting in larger regurgitant orifice in COAPT. This is a fact that needs no discussion and is undoubtedly a key element.
LV Dimensions
The finding of larger left ventricular (LV) volumes in MITRA-FR has been noted as a very important difference between the two studies. This is likely, but it is not as evident as it is usually presented. LV volumes, assessed by the Simpson method in both studies, matched well, with regurgitant volume measured using the proximal isovelocity surface area method in MITRA-FR but not in COAPT. Therefore, larger LV volumes observed in MITRA-FR are possibly due to differences in core lab evaluations (Table 1).
Proportionate/Disproportionate Theory
The concept of proportionate/disproportionate MR—ratio of MR severity/LV dimension and whether the degree of MR is expected or not for the observed degree of LV dilatation—has emerged as a framework to predict which patient might benefit from MR correction.12 The main principle underpinning this concept is that when the degree of MR is significantly higher than what would be expected for the observed degree of LV remodeling (disproportionate), MR is driving the outcome, and consequently, its correction might be beneficial. In contrast, in patients with proportionate MR, LV remodeling/dysfunction is the main determinant of the outcome, so MR correction would not affect the outcome. However, the concept of disproportionate MR is not supported by a recent MITRA-FR post hoc subanalysis, in which patients with disproportionate MR derived no further benefit from the MitraClip (Abbott) (Figure 3).13,14
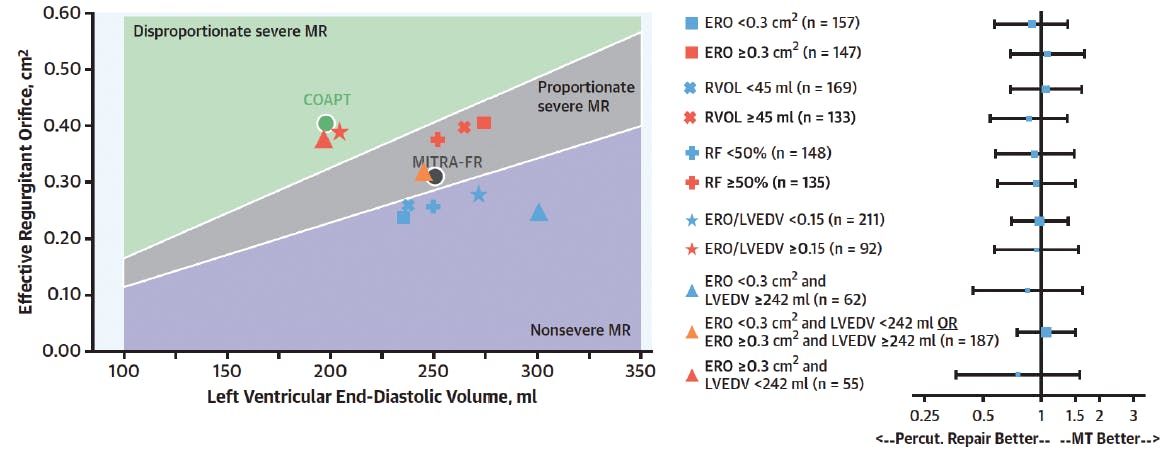
Figure 3. Even in the group with effective regurgitation orifice (ERO) ≥ .3 cm2 and LV < 242 mL (n = 55) or ERO/LV end-diastolic volume ≥ .15 (n = 92), we saw no difference between the groups. Those subgroups are probably too small but the efficacy reported in COAPT is so important that even with those small numbers, we should see at least a trend. This secondary analysis does not support the proportionate/disproportionate concept. LVEDV, left ventricle end-diastolic volume; RF, radiofrequency. Reprinted with permission from Messika-Zeitoun D, Iung B, Armoiry X, et al. Impact of mitral regurgitation severity and left ventricular remodeling on outcome after MitraClip implantation: results from the Mitra-FR Trial. JACC Cardiovasc Imaging. 2021;14:742-752.
As a consequence, the proportionate/disproportionate concept, although appealing, cannot rely on a really objective analysis of the two studies. We generally agree that secondary MR can no longer be defined just by the severity of the regurgitation (ie, effective mitral regurgitant orifice area, regurgitation volume) and that we have to include LV parameters. However, we still do not know which parameters to use (ie, systolic or diastolic volumes, indexed) and how to include the function (ie, ejection fraction, stroke volume, regurgitation fraction). More studies are required to refine this very promising concept.
“COAPT-Like” Patients
This concept has emerged after secondary analysis of the COAPT trial, particularly by echocardiographic selection criteria, which were not mentioned in the initial protocol. It has been popularized after the definition of the FDA approval. In a secondary analysis of MITRA-FR, the subset of “COAPT-eligible” patients was not different between the clip and medical arms (Table 2).15 Restricting patient selection to those who meet the eligibility criteria of COAPT is probably oversimplistic. This is not enough to reproduce the selection imposed by the COAPT eligibility committee and therefore cannot guaranty the same efficacy.
Central Eligibility Committee
Patients enrolled in COAPT were highly selected and validated by an eligibility committee, contrary to those enrolled in MITRA-FR. Therefore, unreported factors such as myocardial reserve, right ventricular function, and severity of the tricuspid regurgitation may also explain the divergent results between the two trials. This committee has likely played a central role in the success of the COAPT trial, and thus it is difficult to compare its results to real life. The MITRA-FR inclusion process was likely more representative of real-life scenarios.
Medical Management
Interpreting medical management is very difficult and leads to more questions than answers. For instance, it is unclear why MR decreased in both study control groups (−46.9% in the COAPT and −32.5% in the MITRA-FR). Guideline-directed medical therapy (GDMT) was compulsory for at least 3 months in both protocols, so patients were supposed to be stabilized at randomization.16,17 After randomization, any modification of medical treatment was discouraged in COAPT, which may have highlighted the difference between the two COAPT groups. Conversely, GDMT was encouraged during the follow-up in MITRA-FR with more than 80% of adoption, meaning that the control group of MITRA-FR respected the GDMT all throughout follow-up. Again, this emphasizes that the MITRA-FR trial was presumably more faithful to real-life practice.
Severity of MITRA-FR Patients
One of the most often cited interpretations of the difference between the two studies is that the MITRA-FR patients were severally ill due to a more advanced disease and too severe LV dysfunction, suggesting that MR was beyond repair. This idea relies on observational studies showing that the prognostic impact of secondary MR is less prominent in patients with advanced heart failure, which suggests that MR correction in these subsets may provide limited benefit.10,18 Unfortunately, this interpretation is also too oversimplistic because it is not supported by the similar ejection fraction in both studies, the higher N-terminal pro–brain natriuretic peptide level in COAPT, and the higher mortality rate in the control group of COAPT (46% vs approximately 34% in the two MITRA-FR groups).
Study Support
MITRA-FR was an academic study with a protocol published 3 years before the final results, whereas COAPT was an industry-driven study with a protocol published 3 months before the final results. Aside from the discussion about the respective advantages and limits of the two supports, we can just report the facts as follows: A meta-analysis of 226 clinical trials showed that an industry study was four times more likely to report a positive outcome (odds ratio [OR], 3.9) and nine times less likely to report an unfavorable event (OR, 0.11).19 Moreover, the two trials shared the same weakness of being open-label studies, which is not marginal because the primary endpoint of the two studies included the decision of rehospitalization for heart failure.
Regulatory and Cost-Effectiveness Implications
The divergent findings between the two trials, still not reconciled, also have regulatory and cost-effectiveness implications. The dramatic positivity reported in COAPT explains the decision by some regulatory agencies to grant approval for percutaneous mitral valve repair (pMVR) in secondary MR. In the absence of a clear view on patients who are most likely to derive a benefit of pMVR, this decision limits the risk to refuse an efficient procedure on a good candidate but it implies a risk of overuse in patients like those included in MITRA-FR.
CONCLUSION
COAPT and MITRA-FR both suggest that pMVR is very safe and also effective in a selective population. Reconciling the two studies is not that easy, and no clear selection criteria can be derived from the two trials. There is a critical need for additional research to identify patient subsets who may benefit from an intervention, but for the time being, we should avoid fast and simple conclusions about this complex disease.
1. Grigioni F, Detaint D, Avierinos J-F, et al. Contribution of ischemic mitral regurgitation to congestive heart failure after myocardial infarction. J Am Coll Cardiol. 2005;45:260-267. doi: 10.1016/j.jacc.2004.10.030
2. Rossi A, Dini FL, Faggiano P, et al. Independent prognostic value of functional mitral regurgitation in patients with heart failure. A quantitative analysis of 1256 patients with ischaemic and non-ischaemic dilated cardiomyopathy. Heart. 2011;97:1675-1680. doi: 10.1136/hrt.2011.225789
3. Michler RE, Smith PK, Parides MK, et al. Two-year outcomes of surgical treatment of moderate ischemic mitral regurgitation. N Engl J Med. 2016;374:1932-1941. doi: 10.1056/NEJMoa1602003
4. Acker MA, Parides MK, Perrault LP, et al. Mitral-valve repair versus replacement for severe ischemic mitral regurgitation. N Engl J Med. 2014;370:23-32. doi: 10.1056/NEJMoa1312808
5. Obadia JF, Messika-Zeitoun D, Leurent G, et al. Percutaneous repair or medical treatment for secondary mitral regurgitation. N Engl J Med. 2018;379:2297-2306. doi: 10.1056/NEJMoa1805374
6. Iung B, Armoiry X, Vahanian A, et al. Percutaneous repair or medical treatment for secondary mitral regurgitation: outcomes at 2 years. Eur J Heart Fail. 2019;21:1619-1627. doi: 10.1002/ejhf.1616
7. Stone GW, Lindenfeld J, Abraham WT, et al. Transcatheter mitral-valve repair in patients with heart failure. N Engl J Med. 2018;379:2307-2318. doi: 10.1056/NEJMoa1806640
8. Magne J, Sénéchal M, Dumesnil JG, et al. Ischemic mitral regurgitation: a complex multifaceted disease. Cardiology. 2009;112:244-59. doi: 10.1159/000151693
9. Grinberg D, Uhlrich W, Thivolet S, et al. The unfinished saga of invasive procedures for secondary mitral regurgitation. Ann Cardiothorac Surg. 2021;10:66-74. doi: 10.21037/acs-2020-mv-15
10. Wu AH, Aaronson KD, Bolling SF, et al. Impact of mitral valve annuloplasty on mortality risk in patients with mitral regurgitation and left ventricular systolic dysfunction. J Am Coll Cardiol. 2005;45:381-387. doi: 10.1016/j.jacc.2004.09.073
11. Acker MA, Parides MK, Perrault LP, et al. Mitral-valve repair versus replacement for severe ischemic mitral regurgitation. N Engl J Med. 2014;370:23-32. doi: 10.1056/NEJMoa1312808
12. Grayburn PA, Sannino A, Packer M. Proportionate and disproportionate functional mitral regurgitation: a new conceptual framework that reconciles the results of the MITRA-FR and COAPT trials. JACC Cardiovasc Imaging. 2018;12:353-362. doi: 10.1016/j.jcmg.2018.11.006
13. Messika-Zeitoun D, Iung B, Armoiry X, et al. Impact of mitral regurgitation severity and left ventricular remodeling on outcome after MitraClip implantation: results from the Mitra-FR trial. JACC Cardiovasc Imaging. 2021;14:742-752. doi: 10.1016/j.jcmg.2020.07.021
14. Obadia JF, Iung B, Messika-Zeitoun D. The disproportionate success of the disproportionate concept. J Thorac Cardiovasc Surg. Published online August 28, 2020. doi: 10.1016/j.jtcvs.2020.06.114
15. Iung B, Messika-Zeitoun D, Boutitie F, et al. Characteristics and outcome of COAPT-eligible patients in the MITRA-FR trial. Circulation. 2020;2482-2484. doi: 10.1161/CIRCULATIONAHA.120.049743
16. Mack MJ, Abraham WT, Lindenfeld JA, et al. Cardiovascular outcomes assessment of the mitraclip in patients with heart failure and secondary mitral regurgitation: design and rationale of the COAPT trial. Am Heart J. 2018;205:1-11. doi: 10.1016/j.ahj.2018.07.021
17. Obadia JF, Armoiry X, Iung B, et al. The MITRA-FR study: Design and rationale of a randomised study of percutaneous mitral valve repair compared with optimal medical management alone for severe secondary mitral regurgitation. EuroIntervention. 2015;10:1354-1360. doi: 10.4244/EIJV10I11A232
18. Goliasch G, Bartko PE, Pavo N, et al. Refining the prognostic impact of functional mitral regurgitation in chronic heart failure. Eur Heart J. 2018;39:39-46. doi: 10.1093/eurheartj/ehx402
19. Riaz H, Raza S, Khan MS, et al. Impact of funding source on clinical trial results including cardiovascular outcome trials. Am J Cardiol. 2015;116:1944-1947. doi: 10.1016/j.amjcard.2015.09.034
Advertisement
Advertisement


