Advertisement
Advertisement
March/April 2025
Comprehensive Multimodality Imaging Assessment of Aortic Regurgitation
A discussion of echocardiographic assessment combined with cardiac MR and CT for comprehensive aortic regurgitation evaluation.
By Takahiro Nishihara, MD; Go Hashimoto, MD; Stephanie Schmidt, RDCS; Paul Sorajja, MD; Vinayak Bapat, MD; Nadira Hamid, MD; Maurice Enriquez-Sarano, MD; and João L. Cavalcante, MD
Aortic regurgitation (AR) is a condition in which the aortic valve lacks coaptation, leading to backward blood flow from the aorta into the left ventricle during diastole and, consequently, progressive volume overload, left ventricular (LV) remodeling, and ultimately dysfunction. A comprehensive assessment of AR severity is crucial for appropriate diagnosis, which leads to proper patient management decision. Although it always involves echocardiography, both cardiac MR (CMR) and CT can provide additive value in the assessment of LV remodeling and AR severity, which will be key to guide management decision. Each modality offers unique advantages and limitations that, when used in conjunction, provide a holistic understanding of the implications of AR.
Evaluation of AR Severity
Echocardiography: Initial Diagnosis and Common Modality
Transthoracic echocardiography (TTE) is the cornerstone for the initial diagnosis of AR. It is a widely available, noninvasive imaging technique that allows real-time visualization of the heart’s structure and function. TTE assesses the characteristics of the AR jets (vena contracta width, ratio of AR jet width/LV outflow tract diameter, eccentricity) using color Doppler imaging; with spectral Doppler, it measures the pressure half-time of the regurgitant jet as well as two-dimensional (2D) proximal isovelocity surface area (PISA). The presence of any holodiastolic flow reversal in the descending or abdominal aorta should prompt further evaluation with transesophageal echocardiography (TEE) or other imaging modalities to confirm the diagnosis and assess the severity and etiology of AR.1 In addition, TTE evaluates LV size and function to gauge the chronic hemodynamic impact of AR.1
However, TTE is not without pitfalls and limitations. Current guidelines emphasize parameters like vena contracta width and regurgitant volume to grade severity, but these measures can vary significantly with loading conditions and operator expertise. Furthermore, eccentric AR jets are common, which can lead to inadequate AR severity assessment, along with inconclusive Doppler data. All of this, along with suboptimal acoustic windows, especially in patients with obesity and/or chronic lung diseases, can lead to inaccurate estimation of AR severity.
TEE: Detailed Diagnostic Insights
TEE often offers enhanced imaging quality with higher spatial and temporal resolution by positioning the ultrasound transducer closer to the aortic valve. This modality is particularly useful in assessing the etiology and mechanism of AR. TEE provides detailed views of the aortic valve leaflets and root, identifying abnormalities such as prolapse, perforation, and structural deformities. In addition, three-dimensional (3D) TEE allows for direct measurement of the vena contracta area, which provides a more accurate assessment of the effective regurgitant orifice area compared to PISA method.2
TEE is invaluable in presurgical planning as it supplies precise anatomic details required for interventions. However, it is semi-invasive and requires the patient to be in a fasting state and under moderate sedation, limiting its use as a routine diagnostic tool compared to TTE.
CMR: Precision in AR Quantification
CMR is the gold standard for quantifying AR due to its high reproducibility and independence from acoustic windows. CMR evaluates aortic regurgitant fraction (RF): the proportion of the aortic regurgitant volume relative to the total stroke volume. A threshold RF of ≥ 33% to 35% typically indicates clinically significant AR.3-5
For optimal results, specific acquisition protocol is necessary. A 2D phase-contrast plane perpendicular to the sinotubular junction, while avoiding aliasing, provides reproducibility and precision for accurate regurgitant volumetric analysis and monitoring of AR severity over time. A typical CMR examination for evaluation of AR severity, LV remodeling, and function can be achieved in most centers in < 20 to 30 minutes. There is no radiation and no need for contrast, unless evaluation of myocardial fibrosis/scarring is warranted. Although MRI availability is not an issue in most countries, access to centers with CMR capabilities remains a concern, which limits the use of this technology. Cardiac implantable electronic devices are no longer a contraindication but can produce artifacts. Additionally, severe patient claustrophobia precludes evaluation.
CT: Anatomic Detailing
Although retrospective electrocardiography-gated functional CTA is used less commonly for AR assessment, it offers superb anatomic delineation of cardiac structures. Multidetector CT (MDCT) can measure the anatomical regurgitant orifice area through multiplanar reconstruction (MPR) techniques, providing insights into the mechanism of AR along with quantification of AR severity (Figure 1). MDCT is particularly useful when TEE or TTE provides equivocal results, when CMR is not available, or when surgical planning necessitates precise anatomic data.
The noninvasive nature of CT and its rapid acquisition make it an appealing option, although it involves ionizing radiation and the use of iodine contrast agents. Hence, CT is often reserved for specific cases in which echocardiography and CMR may not provide sufficient details.
EVALUATION OF LV REMODELING AND FUNCTION
The presence of severe AR is associated with progressive volume overload and pressure overload on the left ventricle. As a compensatory mechanism to maintain forward cardiac output, the left ventricle progressively dilates, which can lead to irreversible LV remodeling and dysfunction. Thus, the timing of intervention prior to the hemodynamic myocardial consequences is important.
2D Echocardiography: Fundamental Evaluation
The 2020 American College of Cardiology/American Heart Association valve guidelines6 show that the parameters for intervention in asymptomatic patients with severe AR are LV ejection fraction (LVEF) ≤ 55%, LV end-systolic dimension (LVESD) > 50 mm, or LVESD index (LVESDi) > 25 mm/m2. However, some studies have reported that an LVESDi ≥ 20 mm/m2 is already associated with increased mortality.7,8
LV Volumes by Echocardiography: A Better Evaluation of LV Remodeling
Basal linear LV dimensions are misleading, particularly because the greatest LV remodeling typically occurs at the midcavity and varies according to patient sex. LV end-systolic volume index (LVESVi) ≥ 45 mL/m2 was independently associated with increased mortality risk in asymptomatic patients with moderately severe to severe AR.8 However, we must pay careful attention to the assessment of LV volumes in patients with poor imaging quality and avoidance of image foreshortening, which reduces reproducibility of this method. Although 3D echocardiography allows for more accurate measurement of LV volumes and LVEF than 2D echocardiography, it still requires good acoustic windows. Furthermore, its incremental role beyond 2D volumes in patients with AR remains to be determined (Figure 2A).
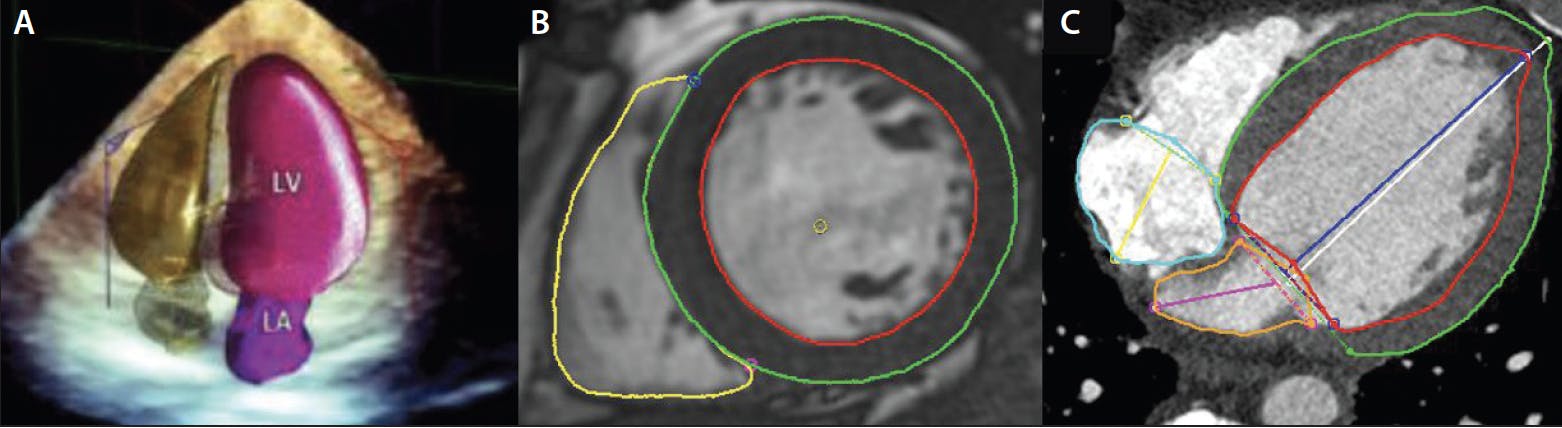
Figure 2. The quantification of LV remodeling by 3D echocardiography (A), cardiac MR (B), and CT (C). LA, left atrium; LV, left ventricle. Reprinted from J Am Soc Echocardiogr, Vol 31, Acquatella H, Asch FM, Barbosa MM, et al, Recommendations for multimodality cardiac imaging in patients with chagas disease: a report from the American Society of Echocardiography in collaboration with the InterAmerican Association of Echocardiography (ECOSIAC) and the cardiovascular imaging department of the Brazilian Society of Cardiology (DIC-SBC), Pages 3-25, Copyright 2018, with permission from Elsevier.
CMR: The Gold Standard Assessment
CMR provides high reproducibility and accuracy for quantification of LV function and volumes (Figure 2B).9 Hashimoto et al suggested that LVESVi > 45 mL/m2 on CMR was associated with increased risk of cardiac events in asymptomatic patients with chronic moderate or severe AR.10 This threshold is similar to the 45 mL/m2 threshold from previous echocardiographic literature.8
Functional CTA: An Alternative Imaging Modality for LV Remodeling and Function
Functional CTA is also useful for the assessment of LV function and LV volume (Figure 2C), offering high accuracy that is comparable to CMR.11 However, the accuracy and quality of these measurements can be affected by the patient’s heart rate, presence of arrhythmia such as atrial fibrillation, and the CT scanner system used for acquisition. Dual-source systems have the best-in-class temporal resolution, allowing for absolute millisecond reconstructions. The technology for volumetric scanners covering the entire heart within one cardiac cycle offers better temporal resolution.
Emerging Parameters
Although LV function and volume analysis relate to outcomes in AR, detecting LV dysfunction in the early stage of the AR journey remains challenging. Therefore, more sensitive indicators of LV remodeling are needed.
Global longitudinal strain (GLS). GLS is a useful parameter to assess myocardial deformation (Figure 3A). Two studies have shown that reduced LV GLS by echocardiography was independently associated with all-cause death in asymptomatic patients with chronic AR and preserved LVEF.12,13 CMR-derived GLS using feature-tracking is a valuable technology and corresponds with clinical outcomes in patients with chronic AR; in a study by Fernández-Golfín et al, the impaired circumferential and radial strain using CMR correlated with AR severity and was associated with increased risk of all-cause mortality, cardiovascular mortality, aortic valve surgery, or cardiovascular hospital admission due to heart failure.14 Further studies to investigate the impact of GLS on the prognosis in patients with AR, including the assessment of CT-derived GLS, are needed.
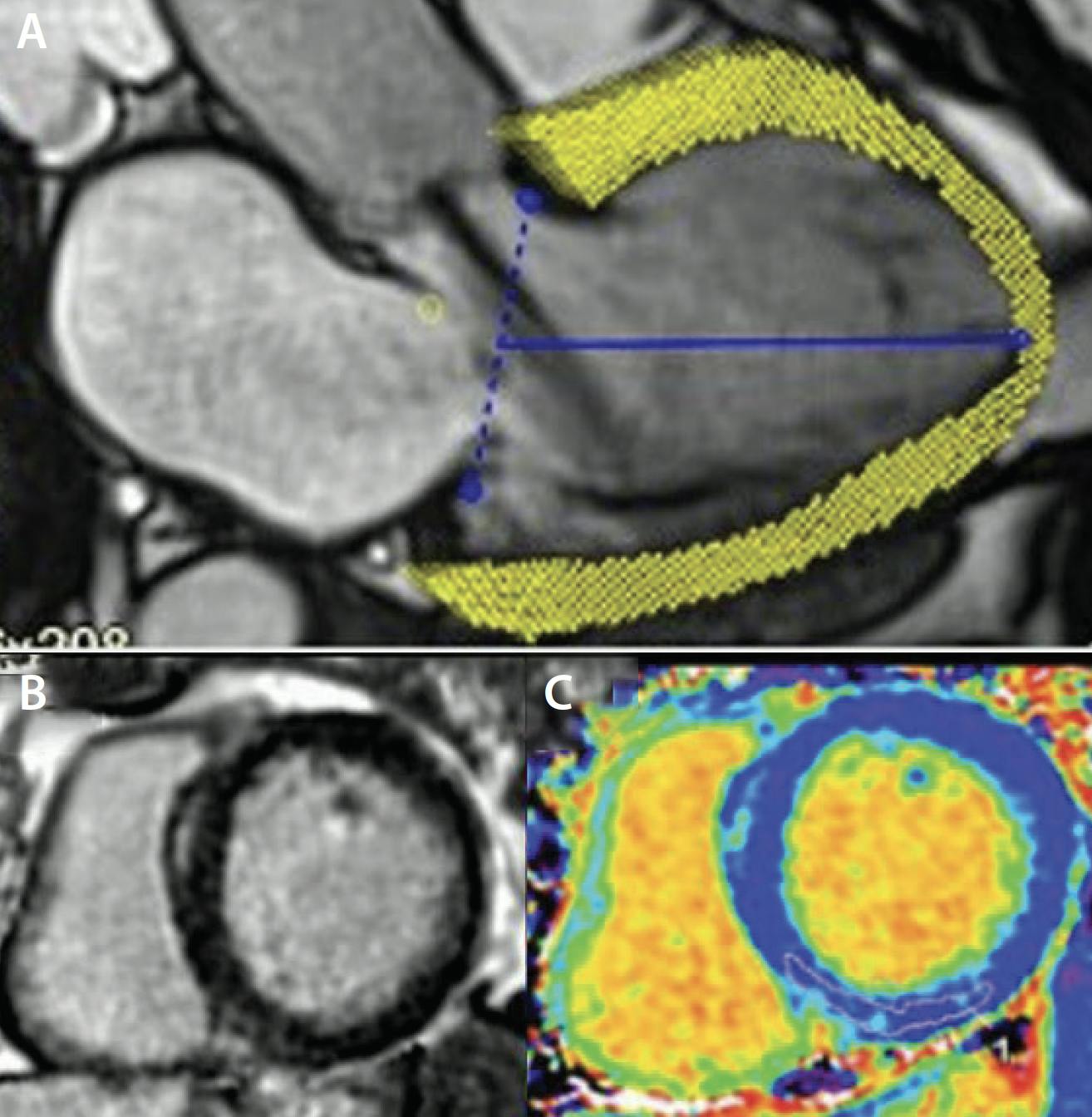
Figure 3. The evaluation of GLS (A), LGE (B), and ECV (C). Reprinted from J Am Coll Cardiol, Vol 82, Ranard LS, Bonow RO, Nishimura R, et al, Imaging methods for evaluation of chronic aortic regurgitation in adults: JACC state-of-the-art review, Pages 1953-1966, Copyright 2023, with permission from Elsevier.
Myocardial fibrosis. CMR and CT are highly effective in detecting and quantifying myocardial fibrosis through techniques such as late gadolinium enhancement (LGE) and extracellular volume (ECV) (Figure 3B). One study found that myocardial scar shown with LGE was present in approximately one-third of patients with AR and was independently associated with a 2.5-fold increase in mortality in patients with moderate or greater AR.15 ECV was significantly increased with AR severity measured by aortic RF and was associated with adverse outcomes in chronic AR patients.16 Cardiac CT can be used as an alternative to evaluate LGE and ECV, but further studies are required.
TRANSCATHETER AORTIC VALVE REPLACEMENT PLANNING
Comprehensive CT Assessment for Transcatheter Aortic Valve Replacement
CT is a valuable tool in the preoperative assessment of patients with AR, providing comprehensive anatomic information about valvular type, presence and pattern of calcification, and evaluation of aorta morphology (Figure 4). The following measurements and imaging features are essential for ensuring accurate valve sizing and optimal prosthesis deployment, as well as minimizing peri- and postprocedural complications17:
1. Aortic annulus dimensions: Annular perimeter and area are critical for selecting the appropriate valve size.
2. Aortic root and ascending aorta: Measurements of the sinus of Valsalva, sinotubular junction, and ascending aorta are essential to ensure proper valve fit and avoid complications such as coronary obstruction.
3. Valve morphology: Assessments of the aortic valve morphology and AR mechanism are important for deployment. Bicuspid aortic valve is a contraindication for current dedicated AR devices.
4. Coronary ostial heights: The distance from the aortic annulus to the coronary ostia must be measured to prevent coronary obstruction during valve deployment.
5. Membranous septum length: This can contribute to increased risk for pacemaker.
6. Aortoiliofemoral system: This is useful for determine the best vascular access route and identifying potential tortuosity, calcification, or pathologies that could cause problems with advancement of the delivery system.
Device-Specific Features
The JenaValve Trilogy (JenaValve Technology, Inc.) and the J-Valve (JC Medical) are both dedicated transcatheter heart valves designed for use in patients with AR, but they have distinct features and anchoring mechanisms that we need to know for CT assessment during transcatheter aortic valve replacement (TAVR) planning. The Trilogy valve consists of a self-expanding nitinol frame with porcine pericardial leaflets. It features locators that clip onto the native leaflets and aid with anchoring.18 The Trilogy valve design features include three large, open cells that facilitate coronary access after implantation, which is crucial for patients who may require future coronary interventions. The Trilogy valve utilizes three device sizes, allowing for treatment of aortic annular perimeters between 66 and 90 mm. Significant aortic root angulation (ie, > 70 degrees) and short annulus to ascending aorta length (ie, < 4-5 cm) can make navigation of the delivery system and alignment more challenging.
The J-Valve device is also a self-expanding nitinol frame with bovine pericardial leaflets.19 It has three U-shaped anchor rings designed to self-orient with the aortic sinuses and then capture the native leaflets. The valve has five different sizes and is designed to treat annular perimeters ranging from 57 to 104 mm.
INTEGRATION OF MODALITIES
Integrating findings from echocardiography, TEE, CMR, and CT enhances diagnostic accuracy and decision-making in AR management. While echocardiography initiates the diagnostic process, misdiagnosis and underestimation of AR severity and LV remodeling often occur, leading to delayed referral to treatment. TEE can provide detailed anatomic assessment of the aortic valve and AR mechanism and, at times, better quantification of AR. CMR provides precise quantification of AR severity and LV remodeling, whereas cardiac CTA can represent a viable option to CMR for both anatomic and functional overview, in addition to serving for preprocedural planning. This comprehensive evaluation strategy represents a paradigm in modern cardiology, aiming to optimize diagnostic precision, management decisions, and therapeutic success.
1. Zoghbi WA, Adams D, Bonow RO, et al. Recommendations for noninvasive evaluation of native valvular regurgitation: a report from the American Society of Echocardiography developed in collaboration with the Society for Cardiovascular Magnetic Resonance. J Am Soc Echocardiogr. 2017;30:303-371. doi: 10.1016/j.echo.2017.01.007
2. Yanagi Y, Kanzaki H, Yonezawa R, et al. Diagnostic value of vena contracta area measurement using three-dimensional transesophageal echocardiography in assessing the severity of aortic regurgitation. Echocardiography. 2021;38:1307-1313. doi: 10.1111/echo.15144
3. Kammerlander AA, Wiesinger M, Duca F, et al. Diagnostic and prognostic utility of cardiac magnetic resonance imaging in aortic regurgitation. JACC Cardiovasc Imaging. 2019;12:1474-1483. doi: 10.1016/j.jcmg.2018.08.036
4. Spampinato RA, Jahnke C, Paetsch I, et al. Grading of aortic regurgitation by cardiovascular magnetic resonance and pulsed Doppler of the left subclavian artery: harmonizing grading scales between imaging modalities. Int J Cardiovasc Imaging. 2020;36:1517-1526. doi: 10.1007/s10554-020-01844-2
5. Postigo A, Pérez-David E, Revilla A, et al. A comparison of the clinical efficacy of echocardiography and magnetic resonance for chronic aortic regurgitation. Eur Heart J Cardiovasc Imaging. 2022;23:392-401. doi: 10.1093/ehjci/jeaa338
6. Otto CM, Nishimura RA, Bonow RO, et al. 2020 ACC/AHA guideline for the management of patients with valvular heart disease: executive summary: a report of the American College of Cardiology/American Heart Association Joint Committee on Clinical Practice Guidelines. Circulation. 2021;143:e35-e71. doi: 10.1161/CIR.0000000000000932
7. Mentias A, Feng K, Alashi A, et al. Long-term outcomes in patients with aortic regurgitation and preserved left ventricular ejection fraction. J Am Coll Cardiol. 2016;68:2144-2153. doi: 10.1016/j.jacc.2016.08.045
8. Yang LT, Anand V, Zambito EI, et al. Association of echocardiographic left ventricular end-systolic volume and volume-derived ejection fraction with outcome in asymptomatic chronic aortic regurgitation. JAMA Cardiol. 2021;6:189-198. doi: 10.1001/jamacardio.2020.5268
9. Grothues F, Smith GC, Moon JC, et al. Comparison of interstudy reproducibility of cardiovascular magnetic resonance with two-dimensional echocardiography in normal subjects and in patients with heart failure or left ventricular hypertrophy. Am J Cardiol. 2002;90:29-34. doi: 10.1016/s0002-9149(02)02381-0
10. Hashimoto G, Enriquez-Sarano M, Stanberry LI, et al. Association of left ventricular remodeling assessment by cardiac magnetic resonance with outcomes in patients with chronic aortic regurgitation. JAMA Cardiol. 2022;7:924-933. doi: 10.1001/jamacardio.2022.2108
11. Asferg C, Usinger L, Kristensen TS, Abdulla J. Accuracy of multi-slice computed tomography for measurement of left ventricular ejection fraction compared with cardiac magnetic resonance imaging and two-dimensional transthoracic echocardiography: a systematic review and meta-analysis. Eur J Radiol. 2012;81:e757-e762. doi: 10.1016/j.ejrad.2012.02.002
12. Alashi A, Mentias A, Abdallah A, et al. Incremental prognostic utility of left ventricular global longitudinal strain in asymptomatic patients with significant chronic aortic regurgitation and preserved left ventricular ejection fraction. JACC Cardiovasc Imaging. 2018;11:673-682. doi: 10.1016/j.jcmg.2017.02.016
13. Alashi A, Khullar T, Mentias A, et al. Long-term outcomes after aortic valve surgery in patients with asymptomatic chronic aortic regurgitation and preserved LVEF: impact of baseline and follow-up global longitudinal strain. JACC Cardiovasc Imaging. 2020;13:12-21. doi: 10.1016/j.jcmg.2018.12.021
14. Fernández-Golfín C, Hinojar-Baydes R, González-Gómez A, et al. Prognostic implications of cardiac magnetic resonance feature tracking derived multidirectional strain in patients with chronic aortic regurgitation. Eur Radiol. 2021;31:5106-5115. doi: 10.1007/s00330-020-07651-6
15. Malahfji M, Senapati A, Tayal B, et al. Myocardial scar and mortality in chronic aortic regurgitation. J Am Heart Assoc. 2020;9:e018731. doi: 10.1161/JAHA.120.018731
16. Senapati A, Malahfji M, Debs D, et al. Regional replacement and diffuse interstitial fibrosis in aortic regurgitation: prognostic implications from cardiac magnetic resonance. JACC Cardiovasc Imaging. 2021;14:2170-182. doi: 10.1016/j.jcmg.2021.04.028
17. Blanke P, Weir-McCall JR, Achenbach S, et al. Computed tomography imaging in the context of transcatheter aortic valve implantation (TAVI)/transcatheter aortic valve replacement (TAVR): an expert consensus document of the Society of Cardiovascular Computed Tomography. JACC Cardiovasc Imaging. 2019;12:1-24. doi: 10.1016/j.jcmg.2018.12.003
18. Adam M, Tamm AR, Wienemann H, et al. Transcatheter aortic valve replacement for isolated aortic regurgitation using a new self-expanding TAVR system. JACC Cardiovasc Interv. 2023;16:1965-1973. doi: 10.1016/j.jcin.2023.07.038
19. Garcia S, Ye J, Webb J, et al. Transcatheter treatment of native aortic valve regurgitation: the North American experience with a novel device. JACC Cardiovasc Interv. 2023;16:1953-1960. doi: 10.1016/j.jcin.2023.05.018
Advertisement
Advertisement


