Advertisement
Advertisement
July/August 2023
Alcohol Septal Ablation
Patient selection, preprocedural planning, a step-by-step procedural overview, and postprocedural management tips for performing septal reduction therapy using alcohol septal ablation in HCM patients with LVOT obstruction.
Left ventricular (LV) outflow tract obstruction is present in nearly 75% of patients with hypertrophic cardiomyopathy (HCM) and may cause significant symptoms. Most patients with obstructive symptoms are successfully managed with medications.1-5 Although effective, there remains a subset of patients who continue to have disabling symptoms despite medications or who are unable to tolerate side effects. Patients who have New York Heart Association class III or IV symptoms with significant gradients and adequate septal thickening are candidates for septal reduction therapy with either myectomy or alcohol septal ablation (ASA).2
The advantages and limitations of both strategies are well known.6 Myectomy is favored in younger (< 65 years) patients at low surgical risk or in patients with additional pathology requiring surgical management. ASA is preferred in older patients with comorbidities that elevate surgical risk. Without a randomized controlled trial (unlikely to occur), it becomes difficult to compare the two strategies because outcomes are influenced by selection bias. Data gleaned from registries and meta-analyses allow some comparisons.7,8 Both procedures reduce gradients and improve symptoms. Myectomy wins over ASA on several fronts, with lower gradients, lower risk of heart block requiring a permanent pacemaker (4% vs 10% for patients undergoing ASA), and lower rate of reintervention (1.6% vs 7.7% for ASA).7,8 It is not surprising that ASA wins over myectomy in terms of shorter length of stay and lower procedural morbidity.9 There is no difference in early (30 day) mortality between the two methods,8 but recent studies derived from large registries suggest there may be a higher, long-term mortality rate in patients undergoing ASA.9,10 Many argue that in these nonrandomized trials, selection bias may strongly influence the results because, not surprisingly, patients treated with ASA were older and had more comorbidities; thus, higher mortality might be due to unaccounted variables rather than a true survival advantage for surgery. There has always been concern that ASA might create a substrate for ventricular arrhythmias that might lead to late arrhythmic events. Although this was not shown in earlier studies,11-13 it might explain a mechanism accounting for a higher mortality with ASA seen in these recent studies. Ultimately, deciding between ASA and myectomy requires a heart team approach and consideration of the risks and benefits of each technique, as well as patient preferences. The ideal patient for ASA meets the criteria for septal reduction stated earlier and is typically older with comorbid conditions that increase the risk for surgery. Septal ablation in healthy young patients is not recommended due to concerns about the late sequelae of the created scar or the concern about having a pacemaker for many years.
PREPROCEDURE PLANNING
A careful review of the echocardiogram should confirm significant obstruction (> 50 mm Hg at rest or with provocation), systolic anterior motion (SAM) of the mitral valve, and “adequate” septal thickening (usually ≥ 1.5 mm). Most importantly, the septal anatomy requires careful review. The ideal anatomy is a proximal first septal perforator from the left anterior descending artery (LAD) that is about 1.5 mm in diameter and reaches not much farther than the midseptum (Figure 1). However, septal anatomy varies greatly, and some varieties are not suitable for ASA. For example, there may be an array of very small septals, or the proximal septals may be too small to instrument or unlikely to supply the correct place in the septum to eliminate obstruction (Figure 2). Alternatively, there may be a single, very large septal perforator that supplies a large amount of nontargeted myocardium (Figure 3). Note that septal perforators may arise from vessels other than the LAD, including the proximal diagonal, ramus, left main, and even right coronary arteries. The operator should review the baseline electrocardiogram. Because a right bundle branch block occurs in 40% of patients undergoing ASA, a pre-existing left bundle branch block is associated with a very high risk for permanent pacemaker.
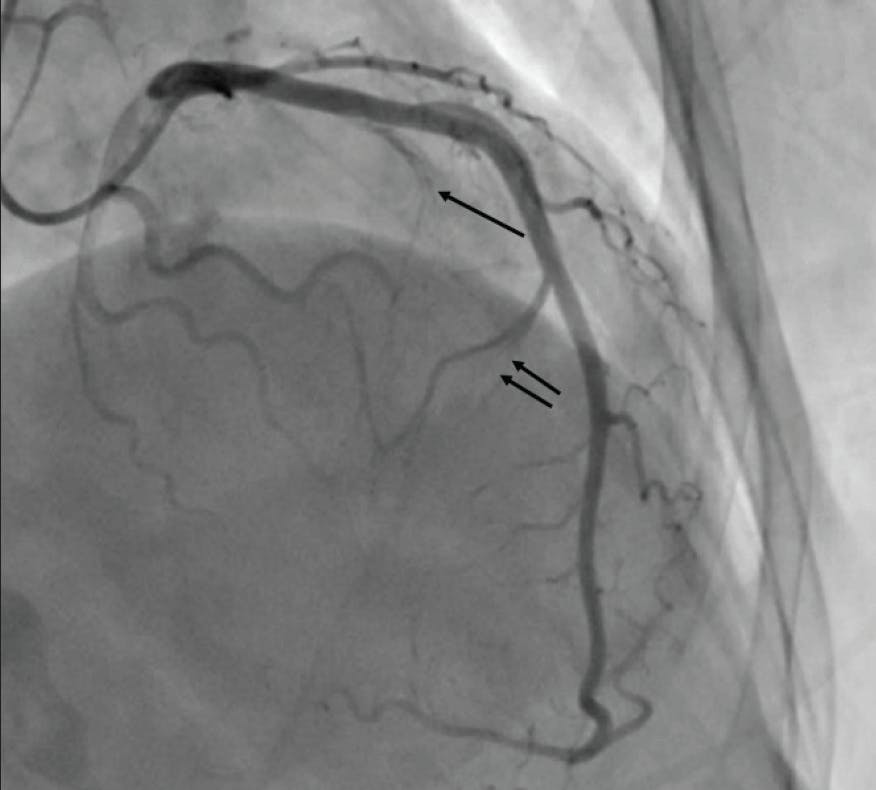
Figure 2. In this patient, the first septal perforator (single arrow) is a very small vessel, and injection with alcohol is unlikely to impact the gradient. A larger second septal perforator (double arrow) supplies the midseptum distal to the SAM contact point, and injection in this vessel will not impact the gradient.
During the informed consent process, the operator should specifically note the 10% risk of permanent pacemaker (> 40% if there is a pre-existing left bundle branch block), rare potential for spillage of alcohol damaging other zones of the heart, arrhythmia, ventricular septal defect (also rare), and possibility of inadequate relief of obstruction. The patient should be informed that alcohol will damage the heart muscle (use the term “heart attack”), and they are likely to experience chest pain up to several hours. It is also important to inform them that, for those without an existing pacemaker, a temporary pacemaker will remain in place up to 48 hours, and they will stay in the hospital for 3 to 4 days after the procedure.
PERFORMING THE PROCEDURE
Detailed descriptions of currently used techniques have been described previously.8,14 Prior to the procedure, a temporary pacemaker suitable to remain in place for up to 48 hours is placed, usually from the right internal jugular vein. Two arterial access sites are obtained: one to perform the ablation and measure aortic pressure and the other to measure LV pressure for assessment of the gradients. Gradients are measured at baseline, and angiography is performed. A short (6-8–mm long) over-the-wire balloon is chosen, with a diameter slightly oversized relative to the diameter of the septal perforator, usually 1.5 to 2 mm. A floppy 0.014-inch angioplasty wire is advanced through the balloon catheter and positioned in the chosen septal perforator. The balloon is advanced into the septal perforator and inflated at low pressure (approximately 4 atm). The wire is withdrawn. Angiography is performed to confirm that the balloon occluded the septal perforator. At this point, the operator may already observe a reduction in the LV aortic pressure gradient.15
A transthoracic echocardiogram is obtained during balloon occlusion, and 0.5 to 1 mL of either angiographic contrast or echo contrast is injected while echo images are obtained. The parasternal long axis and apical two-chamber views are ideal for demonstrating the septum and the SAM contact point. Patients with ideal septal anatomy demonstrate a bright contrast effect in the proximal septum at the point of SAM contact (Figure 4). It is important to be sure that the septal perforator does not also supply other areas of myocardium, such as the inferior wall not targeted for ablation. If the anatomy is appropriate to continue, it is confirmed that balloon remains occlusive by injecting contrast into the left coronary artery before proceeding with alcohol injection.
Denatured alcohol is injected (over 1-3 minutes) through the lumen of the inflated balloon into the septal perforator and allowed to dwell for 5 to 10 minutes. The optimal dose varies depending on the size of the septal perforator, thickness of the myocardium, and degree of obstruction. Early in the experience, some operators used a fixed dose of alcohol as high as 5 mL; however, most experts currently recommend lower doses of alcohol to reduce the risk of heart block. The dose should be tailored to the patient, and dose selection is somewhat of an art form by an operator experienced with the procedure. The optimal balance between achieving adequate gradient reduction and lowest risk of heart block is a dose of 1.5 to 2.5 mL.8,16 Representative hemodynamic effects obtained from ASA are shown in Figure 5. The effect on hemodynamics is seen immediately. Successful procedures reduce the gradient by at least 50%. If the observed effect is believed to be inadequate, additional alcohol may be injected in adjacent septal perforators, again weighing the risk of heart block to the benefit of gradient reduction. Postprocedure angiography typically shows occlusion of the targeted septal perforator (Figure 6).
POSTPROCEDURE MANAGEMENT
Patients are often admitted to an intensive care unit with the temporary pacemaker kept in place. Approximately 40% of patients develop right bundle branch block, and usually there is evidence of septal infarction on the electrocardiogram. Transient complete heart block occurs in the cardiac catheterization laboratory in approximately 14% of patients. Complete heart block recurs or persists in 44% of these patients, and most within 48 hours of the procedure.17 Thus, if after 48 hours there is no persistence or recurrence of complete heart block, the temporary system may be removed. For patients without a permanent pacemaker, monitoring should be continued in the hospital an additional 24 to 48 hours as it is possible to develop heart block out to 72 hours after ASA17; heart block after 4 to 5 days is rare. Some advocate for measuring cardiac enzymes after the procedure, but it is not clear whether this is helpful in decision-making. It is also usually not helpful to obtain an echocardiogram in the hospital as the effect on the outflow tract gradient generally continues to improve over time.18 Medications for HCM are typically continued until patients are seen at follow-up. Repeat echocardiography is often performed at 1 to 2 months to assess the effect of ASA.
Recently, transcatheter approaches to replace the mitral valve have been developed. Known as transcatheter mitral valve repair (TMVR), one of the major limitations of these procedures is the potential for LV outflow tract obstruction.19 ASA has recently been applied to patients under consideration for TMVR at high risk for LV outflow tract obstruction.20 These procedures can be effective but are also challenging because, unlike patients with HCM, the septum may not be abnormally thick, and the septal arteries may be small in caliber. There is a paucity of data regarding the safety and effectiveness of this procedure, and there is significant concern about creating a ventricular septal defect, particularly if the septum is < 1.2 cm. Typically, no more than 1 mL of denatured alcohol is needed for this indication.
CONCLUSION
ASA can be an effective therapy in symptomatic patients with LV outflow tract obstruction from HCM and suitable anatomy. Optimal results rely on careful preprocedural planning and the procedure is guided by hemodynamic assessment and contrast echocardiography.
1. Maron BJ, Desai MY, Nishimura RA, et al. Diagnosis and evaluation of hypertrophic cardiomyopathy. JACC State-of-the-Art Review. J Am Coll Cardiol. 2022;79:372-389. doi: 10.1016/j.jacc.2021.12.002
2. Ommen S, Mital S, Burke M, et al. 2020 AHA/ACC guideline for the diagnosis and treatment of patients with hypertrophic cardiomyopathy: a report of the American College of Cardiology/American Heart Association Joint Committee on Clinical Practice Guidelines. J Am Coll Cardiol. 2020;76:e159-240. doi: 10.1016/j.jacc.2020.08.045
3. Maron BJ. Clinical course and management of hypertrophic cardiomyopathy. N Engl J Med. 2018;379:655-668. doi: 10.1056/NEJMra1710575
4. Wheeler MT, Olivotto I, Elliott PM, et al. Effects of mavacamten on measures of cardiopulmonary exercise testing beyond peak oxygen consumption. A secondary analysis of the EXPLORER-HCM randomized trial. JAMA Cardiol. 2023;8:240-247. doi: 10.1001/jamacardio.2022.5099
5. Olivotto I, Oreziak A, Barriales-Villa R, et al. EXPLORER-HCM study investigators. Mavacamten for treatment of symptomatic obstructive hypertrophic cardiomyopathy (EXPLORER-HCM): a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet. 2020;396:759-769. doi: 10.1016/S0140-6736(20)31792-X
6. Sorajja P. Alcohol septal ablation for obstructive hypertrophic cardiomyopathy. A word of balance. J Am Coll Cardiol. 2017;70:489-494. doi: 10.1016/j.jacc.2017.06.011
7. Agarwal S, Tuzcu EM, Desai MY, et al. Updated meta-analysis of septal alcohol ablation versus myectomy for hypertrophic cardiomyopathy. J Am Coll Cardiol. 2010;55:823-834. doi: 10.1016/j.jacc.2009.09.047
8. Liebregts M, Vriesendorp PA, ten Berg JM. Alcohol septal ablation for obstructive hypertrophic cardiomyopathy: a word of endorsement. J Am Coll Cardiol. 2017;70:481-488. doi: 10.1016/j.jacc.2017.02.080
9. Mentias A, Smdira NG, Krishnaswamy A, et al. Survivial after septal reduction in patients >65 years old with obstructive hypertrophic cardiomyopathy. J Am Coll Cardiol. 2023;81:105-115. doi: 10.1016/j.jacc.2022.10.027
10. Cui H, Schaff HV, Wang S, et al. Survival following alcohol septal ablation or septal myectomy for patients with obstructive hypertrophic cardiomyopathy. J Am Coll Cardiol. 2022;79:1647-1655. doi: 10.1016/j.jacc.2022.02.032
11. Steggerda RC, Damman K, Balt JC, et al. Periprocedural complications and long-term outcome after alcohol septal ablation versus surgical myectomy in hypertrophic obstructive cardiomyopathy: a single center experience. JACC Cardiovasc Interv. 2014;7:1227-1234. doi: 10.1016/j.jcin.2014.05.023
12. Kwon DH, Kapadia SR, Tuzcu EM, et al. Long term outcomes in high-risk symptomatic patients with hypertrophic cardiomyopathy undergoing alcohol septal ablation. JACC Cardiovasc Interv. 2008;1:432-438. doi: 10.1016/j.jcin.2008.05.009
13. Liebregts M, Steggerda RC, Vriesendorp PA, et al. Long-term outcome of alcohol septal ablation for obstructive hypertrophic cardiomyopathy in the young and the elderly. JACC Cardiovasc Interv. 2016;9:463-469. doi: 10.1016/j.jcin.2015.11.036
14. Holmes DR, Valeti US, Nishimura RA. Alcohol septal ablation for hypertrophic cardiomyopathy: indications and technique. Catheter Cardiovasc Interv. 2005;66:375-389. doi: 10.1002/ccd.20500
15. Almasood A, Garceau P, Woo A, et al. Time to significant gradient reduction following septal balloon occlusion predicts the magnitude of final gradient response during alcohol septal ablation in patients with hypertrophic obstructive cardiomyopathy. JACC Cardiovasc Interv. 2011;4:1030-1034. doi: 10.1016/j.jcin.2011.06.008
16. Kimmelstiel C, Rowin EJ. Fixed, high-volume alcohol dose for septal ablation: high risk with no benefit. Catheter Cardiovasc Interv. 2020;95:1219-1220. doi: 10.1002/ccd.28917
17. El-Sabawi B, Nishimura RA, Cha YM, et al. Transient complete heart block after alcohol septal ablation. Incidence of recurrence and need for permanent pacemaker. Circulation: Cardiovasc Interv. 2020;13:e009202. doi: 10.1161/CIRCINTERVENTIONS.120.009292
18. Fernandes VL, Nagueh SF, Franklin J, et al. A prospective follow-up of alcohol septal ablation for symptomatic hypertrophic obstructive cardiomyopathy—The Baylor experience (1996–2002). Clin Cardiol. 2005;28:124-130. doi: 10.1002/clc.4960280305
19. Yoon SH, Bleiziffer S, Latib A, et al. Predictors of left ventricular outflow tract obstruction after transcatheter mitral valve replacement. J Am Coll Cardiol. 2019;12:182-193. doi: 10.1016/j.jcin.2018.12.001
20. Elhadi M, Guerrero M, Collins JD, et al. Safety and outcomes of alcohol septal ablation prior to transcatheter mitral valve replacement. JSCAI. 2022;1:100396. doi: 10.1016/j.jscai.2022.100396
Advertisement
Advertisement





