Advertisement
Advertisement
May/June 2021
Pullback Pressure Gradient: Discriminating Focal and Diffuse Coronary Artery Disease Using Coronary Physiology
Improving post-PCI clinical outcomes.
By Daniel Munhoz, MD, PhD; Jeroen Sonck, MD; and Carlos Collet, MD, PhD
Stable coronary artery disease (CAD) has been studied until now as a single disease entity. The severity of CAD is assessed by the presence and extension of ischemia.1,2 Myocardial ischemia has been proposed as the end mechanism leading to adverse clinical events.3 Percutaneous coronary intervention (PCI) is an effective tool to treat patients with ischemic heart disease. PCI restores epicardial conductance, improves myocardial perfusion, relieve patients from angina, and reduces myocardial infarction.4-6
Randomized controlled trials have established the benefit of invasive functional assessment to guide clinical decision-making about myocardial revascularization in patients with stable CAD.7,8 Fractional flow reserve (FFR) is a vessel-level metric surrogate of myocardial ischemia.2 Beyond determining the hemodynamic significance, coronary physiology can be used to classify CAD further into two disease phenotypes, focal and diffuse disease.
The characterization of focal or diffuse epicardial disease has been commonly done using conventional angiography. Based on coronary angiograms, different definitions have been proposed aiming at differentiating diffuse and focal CAD.9,10 However, assessing the pattern of atherosclerosis using angiography is often equivocal and has low interobserver reproducibility.11 Moreover, studies using intravascular ultrasound have demonstrated diffuse atherosclerosis even in normal angiographic segments.12,13 An alternative approach to estimate the degree of diffuseness of CAD is coronary physiology using intracoronary pressure pullbacks. Pullback curves depict the distribution of epicardial resistance and expose the functional pattern of CAD. Furthermore, a pullback maneuver identifies the location of pressure step-ups in the coronary vessel as targets for PCI (Figure 1).14
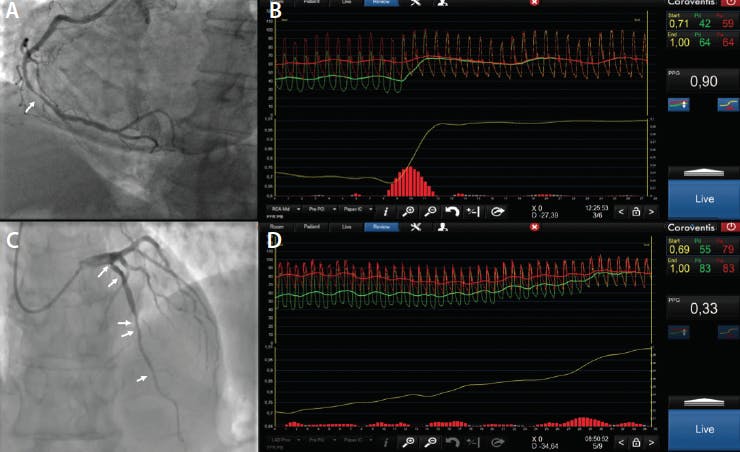
Figure 1. Morphologic and functional CAD pattern evaluation. Coronary angiography showing a right coronary artery (RCA) with angiographically focal lesion (white arrow) (A). FFR pullback of the RCA showing a focal pattern of CAD with a PPG of 0.90 (B). Coronary angiography showing a left anterior descendent artery (LAD) with diffuse lesions (white arrows) (C). FFR pullback of the LAD showing a diffuse pattern with a PPG of 0.33 (D).
PULLBACK PRESSURE GRADIENT INDEX
The discrepancy between anatomy and physiology for lesion significance has been widely recognized.15,16 This includes the evaluation of the pattern of CAD as either focal or diffuse. Focal angiographic disease can exhibit a diffuse pattern of pressure losses along the coronary vessel, and conversely, patients with diffuse angiographic CAD can present with focal pressure losses (Figure 2).14 Until now, the assessment of pressure pullback curves relied on visual assessment. The pullback pressure gradient (PPG) is a novel metric that quantifies the pattern of CAD based on FFR pullbacks. The PPG incorporates the magnitude and extension of pressure losses, providing a continuous metric from 0 to 1; values close to 0 represent diffuse CAD, whereas values close to 1 represent focal CAD.14
Differentiating patients with hemodynamically significant lesions into the two disease phenotypes portends clinical and therapeutic implications. Patients with focal disease have a more severe reduction in myocardial perfusion, lower FFR, and higher transstenotic gradients.14 These features have been associated with plaque vulnerability and a worse prognosis.17 Conversely, patients with diffuse disease have relatively higher FFR values and lower plaque stress.17 Furthermore, treatment options differ in their ability to reestablish epicardial conductance.18 In focal CAD, PCI restores epicardial conductance and relieves ischemia.19 In contrast, PCI in cases of diffuse CAD results in a minor improvement in epicardial conductance and low post-PCI FFR.20 It can be hypothesized that patients with a high PPG (or focal CAD) benefit the most from PCI. The PPG is the first metric that enables this discrimination of CAD patterns. It provides a new opportunity to improve the management of patients with CAD considered for revascularization.
CLINICAL IMPLICATIONS
In clinical practice, after a successful angiographic PCI, one-third of patients remain with a suboptimal post-PCI FFR.21 Low post-PCI FFR has been associated with an increased risk of adverse events and cardiac death.8,22,23 The distribution of coronary atherosclerosis at baseline (eg, focal or diffuse) is a major determinant of the success of PCI in terms of coronary physiology. The calculation of the PPG is indicated in vessels with significant hemodynamic lesions. PPG is performed using a manual pullback for 20 to 30 seconds. We’ve shared details on how to perform a manual FFR pullback maneuver and calculate the PPG in the catheterization video online.24 Further characterization of hemodynamically significant lesions into two different phenotypes aids in improved patient selection for PCI and anticipating PCI benefits. The PPG personalizes clinical decision-making and provides a prediction of post-PCI FFR before the intervention.
The adoption of coronary physiology in clinical practice continues to increase, substantiated by the evidence of clinical benefit of FFR-guided revascularization strategy.1 The current approach that relies on detecting hemodynamic significance of epicardial lesions will be further enriched by discriminating functional CAD phenotypes and personalizing management. Coronary physiology expands from a diagnostic instrument to detect hemodynamically significant lesions to a planning tool for revascularization. PPG has been shown to predict the degree of functional revascularization, bearing the potential to enhance patient selection for PCI and improve clinical outcomes. Higher PPG at baseline results in higher post-PCI FFR. Conversely, PCI in patients with a low PPG results in lower post-PCI FFR (Figure 3).20 Furthermore, patients with a high PPG are often free from angina after the procedure, whereas patients with a low PPG have a higher rate of recurrent angina after PCI.25 It can be hypothesized that patients with a high PPG have a better clinical prognosis than patients with a low PPG. This hypothesis is being tested in the ongoing prospective evaluation of the impact of the PPG index on clinical decision-making and outcomes in the PPG Global Registry (NCT04789317).

Figure 3. Interaction between the CAD pattern and PCI. FFR of 0.71 with a pre-PCI FFR pullback of focal CAD with a PPG of 0.90 (A). Post-PCI FFR pullback with distal FFR of 0.96 showing no residual disease (B). FFR of 0.71 with pre-PCI FFR pullback of diffuse CAD with schematic representation—PPG of 0.33 (C). Post-PCI FFR pullback with distal FFR of 0.81 with treated diffuse CAD showing residual diffuse disease (D).
Prediction of functional revascularization represents the next frontier in interventional cardiology. The PPG adds a new dimension in the evaluation of patients considered for revascularization, enhancing clinical decision-making and tailoring the revascularization strategy. Future clinical trials will provide further insights on the value of a PPG-guided PCI strategy on clinical outcomes.
1. Knuuti J, Wijns W, Saraste A, et al. 2019 ESC Guidelines for the diagnosis and management of chronic coronary syndromes. Eur Heart J. 2020;41:407-477. doi: 10.1093/eurheartj/ehz425
2. Pijls NH, De Bruyne B, Peels K, et al. Measurement of fractional flow reserve to assess the functional severity of coronary-artery stenoses. N Engl J Med. 1996;334:1703-1708. doi: 10.1056/NEJM199606273342604
3. Maron DJ, Hochman JS, Reynolds HR, et al. Initial invasive or conservative strategy for stable coronary disease. N Engl J Med. 2020;382:1395-1407. doi: 10.1056/NEJMoa1915922
4. Chaitman BR, Alexander KP, Cyr DD, et al. Myocardial infarction in the ISCHEMIA trial: impact of different definitions on incidence, prognosis, and treatment comparisons. Circulation. 2021;143:790-804. doi: 10.1161/CIRCULATIONAHA.120.047987
5. Spertus JA, Jones PG, Maron DJ, et al. Health-status outcomes with invasive or conservative care in coronary disease. N Engl J Med. 2020;382:1408-1419. doi: 10.1056/NEJMoa1916370
6. Shaw LJ, Berman DS, Maron DJ, et al. Optimal medical therapy with or without percutaneous coronary intervention to reduce ischemic burden: results from the Clinical Outcomes Utilizing Revascularization and Aggressive Drug Evaluation (COURAGE) trial nuclear substudy. Circulation. 2008;117:1283-1291. doi: 10.1161/CIRCULATIONAHA.107.743963
7. Tonino PA, De Bruyne B, Pijls NHJ, et al. Fractional flow reserve versus angiography for guiding percutaneous coronary intervention. N Engl J Med. 2009;360:213-224. doi: 10.1056/NEJMoa0807611
8. Xaplanteris P, Fournier S, Pijls NHJ, et al. Five-year outcomes with PCI guided by fractional flow reserve. N Engl J Med. 2018;379:250-259. doi: 10.1056/NEJMoa1803538
9. Sianos G, Morel MA, Kappetein AP, et al. The SYNTAX score: an angiographic tool grading the complexity of coronary artery disease. EuroIntervention. 2005;1:219-227.
10. Shiono Y, Kubo T, Honda K, et al. Impact of functional focal versus diffuse coronary artery disease on bypass graft patency. Int J Cardiol. 2016;222:16-21. doi: 10.1016/j.ijcard.2016.07.052
11. Zir LM, Miller SW, Dinsmore RE, et al. Interobserver variability in coronary angiography. Circulation. 1976;53:627-632. doi: 10.1161/01.cir.53.4.627
12. Mintz GS, Painter JA, Pichard AD, et al. Atherosclerosis in angiographically “normal” coronary artery reference segments: an intravascular ultrasound study with clinical correlations. J Am Coll Cardiol. 1995;25:1479-1485. doi: 10.1016/0735-1097(95)00088-l
13. De Bruyne B, Pijls NH, Smith L, et al. Coronary thermodilution to assess flow reserve: experimental validation. Circulation. 2001;104:2003-2006. doi: 10.1161/hc4201.099223
14. Collet C, Sonck J, Vandeloo B, et al. Measurement of hyperemic pullback pressure gradients to characterize patterns of coronary atherosclerosis. J Am Coll Cardiol. 2019;74:1772-1784. doi: 10.1016/j.jacc.2019.07.072
15. De Bruyne B, Pijls NHJ, Kalesan B, et al. Fractional flow reserve-guided PCI versus medical therapy in stable coronary disease. N Engl J Med. 2012;367:991-1001. doi: 10.1056/NEJMoa1205361
16. Toth G, Hamilos M, Pyxaras S, et al. Evolving concepts of angiogram: fractional flow reserve discordances in 4000 coronary stenoses. Eur Heart J. 2014;35:2831-2838. doi: 10.1093/eurheartj/ehu094
17. Lee JM, Choi G, Koo BK, et al. Identification of high-risk plaques destined to cause acute coronary syndrome using coronary computed tomographic angiography and computational fluid dynamics. JACC Cardiovasc Imaging. 2019;12:1032-1043. doi: 10.1016/j.jcmg.2018.01.023
18. Gould KL, Nakagawa Y, Nakagawa K, et al. Frequency and clinical implications of fluid dynamically significant diffuse coronary artery disease manifest as graded, longitudinal, base-to-apex myocardial perfusion abnormalities by noninvasive positron emission tomography. Circulation. 2000;101:1931-1939. doi: 10.1161/01.cir.101.16.1931
19. Baranauskas A, Peace A, Kibarskis A, et al. FFR result post PCI is suboptimal in long diffuse coronary artery disease. EuroIntervention. 2016;12:1473-1480. doi: 10.4244/EIJ-D-15-00514
20. Sonck J. Clinical validation of a virtual coronary interventions planner. Presented at: Euro PCR 2021; May 18-20, 2021; virtual presentation.
21. Agarwal SK, Kasula S, Hacioglu Y, et al. Utilizing post-intervention fractional flow reserve to optimize acute results and the relationship to long-term outcomes. JACC Cardiovasc Interv. 2016;9:1022-1031. doi: 10.1016/j.jcin.2016.01.046
22. Piroth Z, Toth GG, Tonino PAL, et al. Prognostic value of fractional flow reserve measured immediately after drug-eluting stent implantation. Circ Cardiovasc Interv. 2017;10:e005233. doi: 10.1161/CIRCINTERVENTIONS.116.005233
23. Lee JM, Hwang D, Choi KH, et al. Prognostic implications of relative increase and final fractional flow reserve in patients with stent implantation. JACC Cardiovasc Interv. 2018;11:2099-2109. doi: 10.1016/j.jcin.2018.07.031
24. CoreAalst. PPG Procedure. Accessed May 6, 2021. https://www.youtube.com/watch?v=KOqKQ4GzSis
25. Collet C. Capacity of the PPG to predict functional revascularisation. Presented at: Euro PCR 2021; May 18-20, 2021; virtual presentation.
Advertisement
Advertisement

