Advertisement
Advertisement
September/October 2016
Sheathless Guide Catheters During Transradial PCI
Bigger catheters in smaller spaces.
Sheathless technology has been part of transradial procedures since the 1940s, when Sig Radner first performed transradial catheterization via radial cutdowns. Migration to femoral vascular access by the modified Seldinger approach using sheaths eclipsed the sheathless approach, which subsequently became lost in the fog of history. The transradial era reinvigorated the need for equipment optimized for the limited vascular space found in many radial arteries, and a variety of potential solutions to maximize working space for interventions are now maturing into the marketplace.
WHY BOTHER WITH SHEATHLESS TECHNOLOGY?
The sheathless approach to radial access avoids the added mechanical stretching of the vascular sheath that is usually on the order of 1 to 2 F larger than the catheter that is passing within it. For instance, a 5-F vascular sheath allows a 5-F catheter to pass within it but is actually around 6 F when its outer diameter is considered. This mechanical effect influences the distal radial artery where its diameter is smallest and limits the size of catheters that can be placed within the sheath and radial artery. Without an external sheath, a 7.5-F guiding catheter has an outer diameter that is still smaller than a typical 6-F vascular sheath, yet it carries the added possibilities of more complex procedures than might be accomplished with a standard 6-F guiding catheter.
POTENTIAL APPROACHES TO SHEATHLESS TECHNOLOGY
In general, industry has been slow to see the potential of transradial access, and a variety of homemade solutions have appeared in the literature that serve to illustrate some design challenges of sheathless technology. The first challenge is forming an interface between the wire placed in the radial artery and the catheter running up the wire to transition smoothly and not razor the vessel from the sharp edge of a guiding catheter tip. Razor effects, as shown in Figure 1, can perforate arteries or core out the endothelial layers resulting in hematoma, radial damage, or even embolization of endothelial material.1 Initial solutions included various mother/daughter combinations where a smaller catheter, typically a multipurpose design, is placed within the guiding catheter to limit differences in size and give a stepwise dilation to catheter passage (Figure 2). Although homemade devices have been successfully used and only require readily available cath lab equipment, the interfaces between catheters are not perfect, and razor effects are potentially still present. A second problem with this approach is that standard catheters developed for femoral procedures are often not coated with lubricious surfaces to minimize friction. This problem is best exemplified by pulling a catheter between your fingers and noting the heat generated by a typical catheter. There are workarounds for reducing catheter friction for uncoated catheters, including using a very wet surface at the point of skin entry or coating the catheter with lubricous vascular fluids, such as ViperSlide (Cardiovascular Systems, Inc.) or RotaGlide (Boston Scientific Corporation), to reduce interface friction with the skin and perivascular tissue.
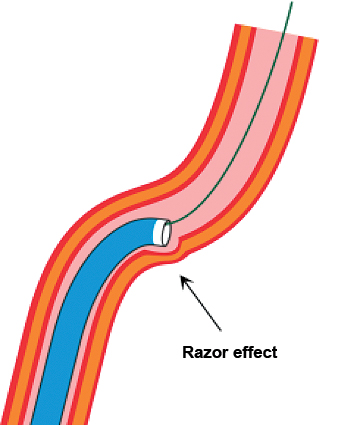
Figure 1. The razor effect. Trauma induced by a mismatch between the inner diameter of catheter and the outer diameter of wire leaves a relatively sharp leading edge of the catheter exposed that may injure vascular structures. Reproduced with permission from Patel T, Shah S, Pancholy S, et al. Working through challenges of subclavian, innominate and aortic arch regions during transradial approach. Catheter Cardiovasc Interv. 2014;84:224–235.

Figure 2. A sheathless system made from a 7-F JR guide catheter with a 5-F multipurpose diagnostic catheter inserted as a dilator, all of which passes over a 0.035-inch wire. The interfaces between the wire and the 5-F catheter and the 5-F catheter and 7-F guide each involve step-like transitions that can induce razor injury on passage through the arterial system. Reproduced with permission from Balahi NR, Resnic FS. Radial catheter selection. Cardiac Interv Today. 2011;5:47–51.
Another ingenuous solution to make standard catheters into sheathless systems involves using an angioplasty balloon protruding from the guiding catheter to act as a smooth interface with the entry site.2 This technique is an extension of the balloon-assisted tracking technique used to advance guides up difficult arteries. In such cases, a balloon appropriately sized to the inner lumen of the guide is inflated as it protrudes from the guide. The assembly is then advanced over a previously placed wire (sized to accept the angioplasty balloon), which is then used as a rail to allow passage through the skin and into the radial artery. Although this provides a nice interface between the guide and wire without razor effects, this approach does not resolve the problem of friction between the guide and perivascular tissue, which needs to be addressed similarly to the mother/daughter-type techniques.
INDUSTRY DEVELOPMENT IN THE SHEATHLESS UNIVERSE
Industry has started to supply some solutions to improve sheathless access. Three general approaches have been suggested and used in various devices worldwide, although not all are available in the United States. The first approach comprises a complete standalone system for vascular access that incorporates both an introducer and a proprietary guide catheter as an integrated device. A second approach uses an introducer that can potentially be placed within many guides and then extends out of the guiding catheter. This approach is designed to reduce or eliminate most of the gap between the introducer and its overriding catheter, yet still potentially leaves open a choice of various guiding catheters. This type of technology has been patented by Cordis Corporation.3 Finally, a third approach involves a nose cone–type configuration that rests over the end of the guiding catheter, which allows a smooth interface between the wire and overriding guide during vascular entry. An example of this is the Flip introducer (Vascular Solutions, Inc.), which is approved in the United States but is not marketed. After the guide is in position in the ascending aorta, the nose cone can be collapsed and removed from the guide when it is no longer needed.
SHEATHLESS EAUCATH CATHETER DESIGN AND USE

Figure 3. The Sheathless Eaucath system with integrated central introducer tailored for minimal clearance over a 0.035-inch wire and interface with the inner lumen of the guiding catheter. The shape of the guiding catheter becomes apparent after the central dilator and wire are removed in the central aorta.
Courtesy of Asahi Intecc Co Ltd
The most developed system on the United States market is the Sheathless Eaucath system (Asahi Intecc Co Ltd.), which provides an integrated introducer and proprietary guiding catheter. The introducer forms a smooth interface between the standard 0.035-inch angiographic wire and its inner core, while the outer surface integrates well with the tip of the guide, essentially eliminating the potential for razor effect (Figure 3). This guiding catheter is available in both 6.5- and 7.5-F sizes with approximately 0.07- and 0.081-inch internal diameters, respectively, and outer diameters slightly smaller than a standard 5- or 6-F sheath, respectively. Similar sheathless technology is available outside of the United States (down to 5-F size; not US Food and Drug Administration approved) from other manufacturers (Medikit Co., Ltd.).
The Sheathless Eaucath system has a hydrophilic coating on the catheter’s exterior that activates when it becomes wet and allows a very smooth entry into the vessel, especially if preceded by a small 4-F sheath. I tend to either place a 4-F radial sheath with a micropuncture kit to initiate the procedure or perform diagnostic angiography to help with the choice of guiding curve. The guide has an internal obturator/dilator with a tapered tip that extends beyond the catheter and passes over a standard 0.035-inch wire. The internal dilator does not have a radiodense tip, and operators need to remain cognizant of its location upon entering the ascending aorta.
Once in the central aorta, the wire and long central core are removed, and the catheter acts like a standard guiding catheter as a function of its predetermined shape, such as standard Judkins or extra-backup type catheters. Given the lubricious nature of the catheter, care needs to be taken to ensure that it does not migrate out of the skin entry site. Use of tape or adhesive transparent medical dressing can anchor the guide at the skin level if needed. Once the catheter at the skin level starts to dry, the hydrophilic coating tends to become somewhat adhesive and helps stabilize the guide at the skin.
At the conclusion of the procedure, the internal core, along with its 0.035-inch wire, is placed back into the guide to prevent the catheter from kinking on withdrawal. The color of the outer surface becomes darker before the guide is fully retracted and can help gauge the location of the tip when preparing for hemostasis and application of hemostasis devices. During this final stage of the procedure, care must be taken not to inadvertently dislocate the guide out of the artery before the hemostasis device is ready. The hydrophilic coating results in very little friction to hold the catheter in the artery, and if it is inadvertently pulled on, it may exit the artery and result in unexpected bleeding. This system is relatively expensive, although there are some cost savings in not requiring a vascular sheath. Likewise, testing multiple guides to find a best fit could become costly with this device.
RESULTS USING SHEATHLESS TECHNOLOGY
Several case series have been reported showing the feasibility of the Sheathless Eaucath system. One potential advantage is the minimization of entry sheath size by eliminating the need for a vascular sheath. This would be expected to result in less vascular entry site complications, such as radial artery occlusion, because these complications are associated with sheath size.4 The hydrophilic coating of the guide that is in contact with the distal radial artery should also lead to less spasm and vascular problems, as suggested in the analogous situation with vascular sheaths.5
Two recent reports using the Sheathless Eaucath system exemplify the experience. The first was a multicenter French registry6 of 148 patients that highlighted a fairly difficult series of percutaneous coronary interventions (PCIs). This patient group included bifurcation lesions (76%), rotational atherectomy (6.1%), thrombus aspiration catheters (12.8%), and chronic total occlusions (6.7%), demonstrating the degree of difficulty. The investigators reported 100% procedural success, with no radial site complications. They concluded that the sheathless system was safe and effective in radial arteries, although they noted the potential for thermal instability of the catheters during long cases, which may represent a future area for catheter improvement. No cases of coronary dissection from guide catheter trauma to the coronary ostia were noted.
The second experience is a Canadian trial in which 233 patients were randomized to PCI with a standard radial vascular sheath and PCI catheters (Launcher, Medtronic) versus the Sheathless Eaucath system.7 Overall, there was similar success as the previously mentioned trial, but the authors noted superior performance by the sheathless system, with greater freedom from crossover to alternative access versus standard guides (96.5% vs 89.9%; P = .047) and less pain perception by patients with the Sheathless Eaucath guides (1.9 ± 1.9 vs 4.8 ± 3.6; P < .001). Beyond this small, randomized experience and the previously mentioned registry from France, a variety of similar case series have shown fairly promising results. Unfortunately, as is often the case with device trials, an appropriately powered randomized trial does not exist for those who demand definitive proof of superiority.
HOMEMADE SHEATHLESS SYSTEMS: RESULTS IN CLINICAL TRIAL
In addition to the new sheathless systems now offered by industry, there are efforts to build sheathless-like devices using preexisting catheters. A recent report from Canada attempted to use a 4-F catheter inside of a 5-F guide as a homemade, sheathless equivalent to pass over a standard angiographic wire up the radial artery.8 Although the step up between the wire and the 4-F catheter followed by the 4-F catheter into the 5-F guide allowed some gradation of dilation into the artery, there were still gaps that potentially could result in razoring of the endothelium and vascular puncture site.
The procedures reported in this series were successful; however, the access sites appeared to have unacceptable damage, which caused the authors to conclude, “Suboptimal transition between diagnostic and guiding catheters likely creates radial artery trauma leading to frequent occlusive thrombus and hematoma surrounding the radial artery.”8 This trial used a relatively small guiding catheter system (4 F in 5 F). Larger systems have also been reported using 7- and 8-F guides. These larger systems might be expected to have a greater tendency for step-up gaps and potential for razor-type injury if studied in larger trials. Although a larger guide may permit a successful intervention, it may come at the cost of increased radial artery damage.
CONCLUSION
The advances being made in sheathless technology are an exciting addition to radial access. Even though only first-generation devices have been studied and data from large-scale studies are lacking, early results suggest a promise of permitting PCI with guides that are less traumatic to the radial artery than the traditional guide-in-sheath approach. Also, there is the potential to allow PCI-sized guides to pass into small arteries in situations such as high radial takeoffs that would not be possible with traditional equipment. Unfortunately, the standalone devices for the sheathless approach may be too expensive (or not available in some circumstances), leaving open the need for understanding homemade sheathless-like systems that may be constructed with standard catheters. Their use comes at the cost of increased radial trauma, but in situations where commercial sheathless technology is not available, this may represent a reasonable compromise when there is a need to minimize the size of the device in the radial artery.
1. Jaworski C, Brown AJ, Hoole SP, West NEJ. From radial artery to embolus: a rare complication of transradial angiography. JACC Cardiovasc Interv. 2015;8:e177-e178.
2. Kwan TW, Ratcliffe JA, Huang Y, et al. Balloon-assisted sheathless transradial intervention (BASTI) using 5 Fr guiding catheters. J Invasive Cardiol. 2012;24:231-233.
3. Sanghvi KA, inventor; Cordis Corporation, assignee. Method and device for sheathless transradial catheterization. WIPO patent 2016/053993A1. April 7, 2016.
4. Uhlemann M, Mobius-Winkler S, Mende M, et al. The Leipzig prospective vascular ultrasound registry in radial artery catheterization: impact of sheath size on vascular complications. JACC Cardiovasc Interv. 2012;5:36-43.
5. Rathore S, Stables RH, Pauriah M, et al. Impact of length and hydrophilic coating of the introducer sheath on radial artery spasm during transradial coronary intervention: a randomized study. JACC Cardiovasc Interv. 2016;3:475-483.
6. Cheaito R, Benamer H, Hovasse T, et al. Feasibility and safety of transradial coronary interventions using a 6.5-F sheathless guiding catheter in patients with small radial arteries. Catheter Cardiovasc Interv. 2015;86:51-58.
7. Noble S, Tessitore E, Gencer B, et al. A randomized study of sheathless vs standard guiding catheters for transradial percutaneous coronary interventions [published online ahead of print April 6, 2016]. Can J Cardiol.
8. Abdelaal E, Rimac G, Plourde G, et al. 4Fr in 5Fr sheathless technique with standard catheters for transradial coronary interventions: technical challenges and persisting issues. Catheter Cardiovasc Interv. 2015;85:809-815.
Ian C. Gilchrist Sr, MD, FACC, FSCAI
Professor of Medicine
Penn State University
College of Medicine
Heart & Vascular Institute
MS Hershey Medical Center
Hershey, Pennsylvania
(717) 531-5888; icg1@psu.edu
Disclosures: None.
Advertisement
Advertisement
