Advertisement
Advertisement
September/October 2016
Radial Approach in Patients With Previous CABG
Data review and tips and tricks.
By Stefano Rigattieri, MD, PhD, and Alessandro Sciahbasi, MD, PhD
In the last 2 decades, a large body of evidence has consistently shown the benefits of the radial artery as a vascular access route for percutaneous diagnostic and interventional procedures.1 Accordingly, the practice of using a radial access (RA) approach has increased worldwide. In 2015, RA was used in nearly 80% of PCIs performed in Italy, whereas in the United States, the rate of transradial PCI increased from 1% in 2007 to 16% in 2012.2
Patients who have undergone previous coronary artery bypass grafting (CABG) represent a challenging subset because they are often older and have a higher prevalence of diabetes and other risk factors as compared to patients who have not undergone CABG. Moreover, peripheral artery disease reflecting more extensive atherosclerotic burden is a common condition3 and could increase the risk of femoral vascular complications, making RA an intriguing option. However, graft PCI is more challenging compared to native coronary artery PCI for several reasons, including variable position of the graft ostia, suboptimal guide catheter support, and the need for adjunctive devices for preventing distal embolization. As compared to the femoral access (FA) approach, RA presents additional technical difficulties, mainly represented by the steeper learning curve, the restriction in terms of catheter size, and the occurrence of radial spasm, which limits multiple catheter exchanges.
In this article, we summarize the available literature data on the use of RA for diagnostic and interventional procedures in patients who have undergone previous CABG. We also discuss some technical tips and tricks and propose a practical flowchart that might help in the selection of vascular access for these patients.
Radial VERSUS Femoral Approach IN CABG PATIENTS: literature data
To date, there is only one randomized study comparing RA to FA in patients with previous CABG.4 The results of this study were clearly in favor of FA, because RA was associated with greater contrast use, longer procedure time, greater access crossover, and greater radiation exposure to the operator compared with FA. Nevertheless, the results of this study cannot be considered conclusive because it was a small, single-center trial, and the experience of the operators was limited (a fellow-first strategy was adopted).
Conflicting results were reported by observational studies performed by expert transradial operators. Similar total procedural times, fluoroscopy times, and contrast volume use were observed by Sanmartin et al in a cohort of patients undergoing diagnostic angiography.5 Rathore et al showed similar rates of success, procedural time, fluoroscopy time, and contrast volume use for RA and FA in a cohort of patients undergoing saphenous vein graft PCI, with a significant reduction in vascular complications in the RA group.6 Similar findings were reported by Bundhoo et al, although they found an increase in radiation dose with RA as compared to FA.7
In a meta-analysis, including the aforementioned studies and amounting to 2,763 patients, we found that RA, as compared to FA, was associated with similar procedural time, fluoroscopy time, procedural success rate, and contrast dose but with a significantly lower rate of vascular complications. However, the main disadvantage of RA was an increased risk of crossover to FA, mainly because of failed selective engagement of the grafts.8
TIPS AND TRICKS FOR SUCCESSFUL RA
1. Ensure that the number and type of conduits are precisely known before starting the procedure. Ignoring the anatomy is associated with increased fluoroscopy time, contrast volume, number of catheters, and access crossover. Of course, knowing the number and location of in situ internal thoracic artery (ITA) grafts is fundamental in the selection of RA.
2. Practice left RA in non-CABG cases, and try to optimize the setup of the cath lab; this will increase your comfort when dealing with CABG patients.
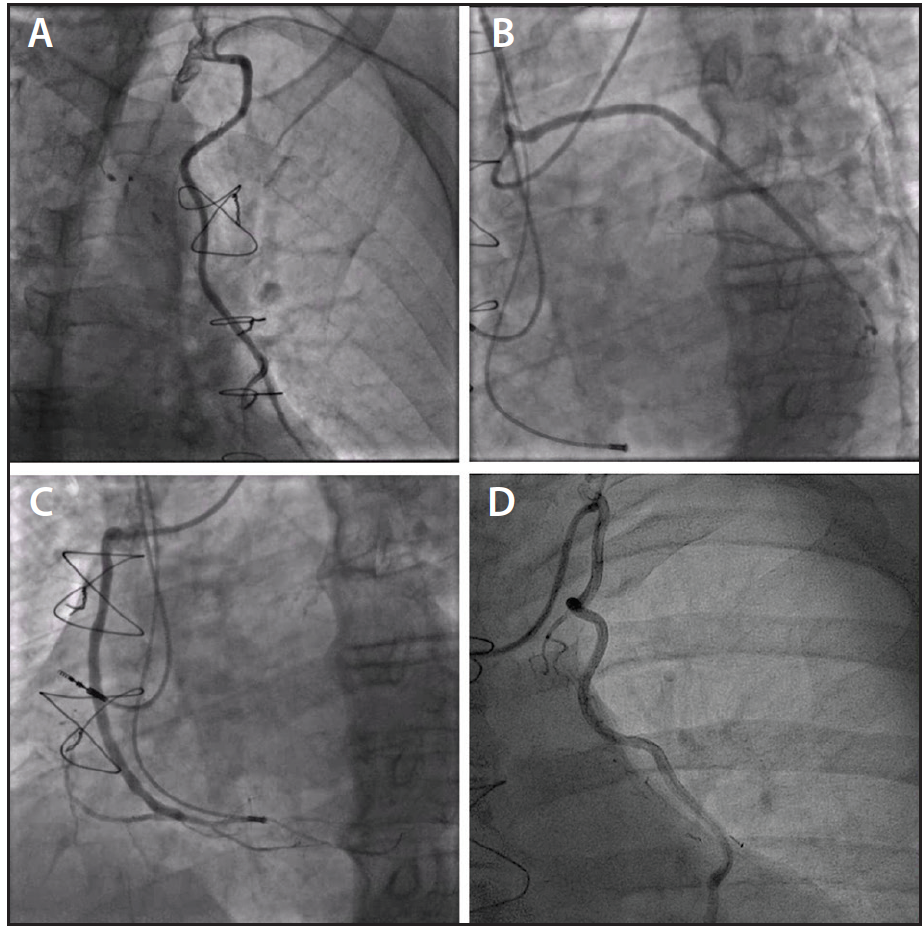
Figure 1. Engagement of the left internal thoracic artery with a Bartorelli-Cozzi catheter via a left radial approach (A). Engagement of a venous graft to the diagonal branch and obtuse marginal branch via a left radial approach (B). Engagement of a venous graft to the right coronary artery via a left radial approach (C). Engagement of the left internal thoracic artery with a mammary catheter via a right radial approach (D).
3. Carefully assess the left radial pulse. Because the artery is frequently used for blood pressure monitoring during surgery, it is useful to assess its patency with the reverse Allen test, in which radial and ulnar arteries are compressed, and the radial is then released to assess hyperemia. Sometimes, the left radial artery is occluded, but you can still appreciate a transmitted pulsation from the palmar arch. In the case of an occluded radial artery, some operators use the homolateral ulnar artery for vascular access.9 This option should be carefully considered, because it carries a risk of hand ischemia in cases of ulnar artery occlusion.
4. Different catheter shapes, both for diagnostic and guiding uses, should be available for left ITA cannulation from the left RA. These include Judkins right, Bartorelli-Cozzi (Figure 1A), internal mammary, and 3D RC (Williams) curves.
5. For diagnostic angiography, a Judkins right catheter is a reasonable first choice for both left and right venous grafts. However, better engagement and support for PCI can be achieved with multipurpose catheters for right grafts and with Amplatz left catheters for left grafts (Figure 1B and 1C).
6. Guide catheter extension systems can be helpful in maximizing support for PCI. Extensions can be used to perform deep intubation or to allow for deep intubation of the guiding catheter (so-called railroading). Alternatively, they can be very useful in maximizing guiding catheter backup with the “swan neck” technique, in which the extension engages the graft while the guiding catheter stands outside and is pushed against the aortic wall.10
7. In the case of radial artery spasm or difficult anatomy, the balloon-assisted tracking technique can be helpful for advancing a guiding catheter on a 0.014-inch coronary guidewire with the help of an inflated 2-mm coronary balloon to allow for a smooth transition avoiding so-called razor effect.11
8. If a guiding catheter larger than 6 F is needed, several options are available: (1) the use of thin-walled sheaths (Slender, Terumo Interventional Systems) that allow the insertion of a 7-F catheter in a 6-F sheath, (2) the use of commercially available sheathless guiding catheters, and (3) the use of homemade sheathless systems consisting of a mother-and-child technique system (5 F in 7 F).
9. Antiembolic protection devices can be easily used with 6-F RAs, because most distal filters are 5-F compatible. A workhorse 0.014-inch buddy wire can be placed in advance to stabilize the system and facilitate filter positioning.
10. Both patient-related and operator-related characteristics play an important role in the selection of vascular access. The presence of clinical conditions that are associated with an increased risk of femoral access complications (eg, obesity, ongoing antithrombotic therapy, and severe peripheral artery disease) should be carefully considered. It is also important to consider risk factors for radial access complications, such as the existence of carotid artery or aortic arch disease or advanced age of the patient (> 75 years), which is associated with a higher prevalence of tortuous subclavian arteries, especially on the right side. The latter conditions may increase the risk for catheter-related cerebrovascular accidents, especially during attempts of ITA engagement coming from the contralateral arm. As far as the operator is concerned, the most important factor is the level of expertise in RA; we suggest that both CABG and primary PCI procedures should be undertaken after having performed at least 150 PCIs in native coronary arteries and in stable patients. A practical flowchart for the selection of vascular access in CABG patients is proposed in Figure 2.
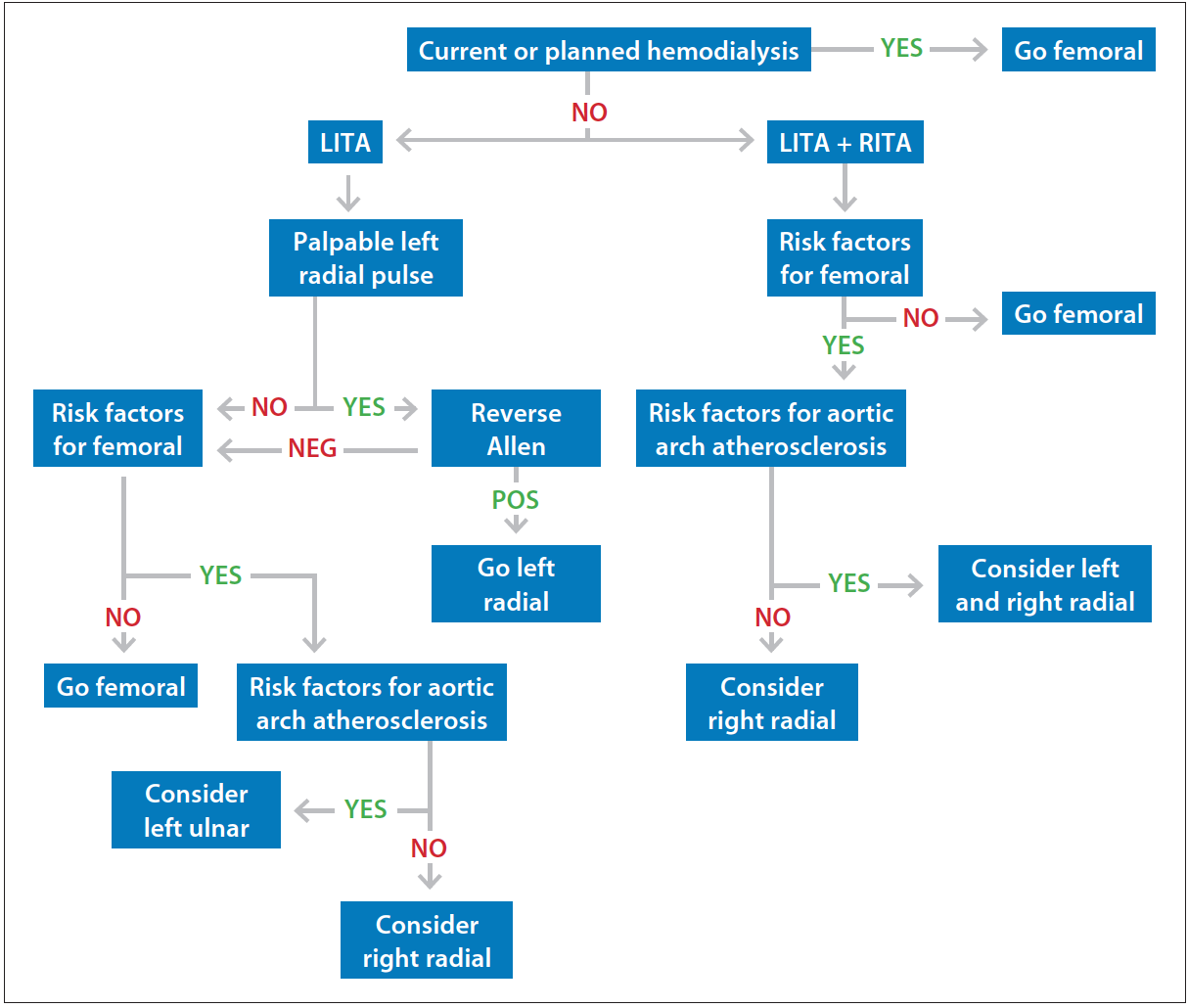
Figure 2. A practical flowchart for the selection of vascular access in CABG patients. Risk factors for a femoral approach include obesity, ongoing antithrombotic therapy, and severe peripheral artery disease. Risk factors for aortic arch atherosclerosis include advanced age (> 75 years) and known carotid or aortic arch disease. Abbreviations: LITA, left internal thoracic artery; RITA, right internal thoracic artery.
WHICH RADIAL ARTERY: LEFT OR RIGHT?
When dealing with native coronary vessels, the right radial artery is by far the preferred approach because the operators are more comfortable given the usual setup of most angiographic suites, with the operator standing at the right side of the patient. However, the scenario is different in most CABG recipients because they have an in situ left ITA graft (usually to the left anterior descending coronary artery) and a variable number of aortocoronary venous grafts.12 In patients with a left ITA graft, the left radial artery should be the access of choice, because the ostium to the left ITA is straight on the way of the catheter path, and the engagement is easy, with good support in case of PCI. The engagement of the left ITA is also feasible using the right radial artery,13 but it requires complex catheter manipulation and is associated with poor catheter stability (Figure 1D). To engage the left ITA coming from a right RA, a left Judkins catheter is advanced in the aortic arch beyond the great vessels and is then slightly pulled back and rotated counterclockwise to point the tip of the catheter toward the origin of the left subclavian artery. Using an exchange-length hydrophilic wire, the subclavian artery is wired, and the left catheter is exchanged for a dedicated catheter to engage the ITA ostium. In the best hands, however, the rate of successful cannulation of the ITA is < 90%.
Although studies comparing left versus right RA for the cannulation of left ITA grafts are lacking, the advantage conferred by left RA is clear; accordingly, left RA was almost exclusively used in studies comparing RA versus FA in patients with in situ left ITA grafts. Similarly, in cases of an in situ right ITA graft, the use of right RA would be preferable to left RA, but in clinical practice, patients with a single right ITA graft are exceedingly rare. It is more frequent to encounter patients with bilateral ITA grafts. In such cases, the use of a single radial approach (usually the right RA because engagement of the contralateral mammary artery is easier from the right RA compared to the left due to the anatomy of the aortic arch) should be reserved for very skilled radial operators because the need for complex catheter manipulation at aortic arch level might increase the risk of cerebral embolization and stroke.
Therefore, in patients with bilateral ITA grafts, two options remain. The first option is the use of FA: in patients at low risk for femoral vascular complications, the use of FA should be preferred because it allows for an easy engagement of both ITAs. The second option is the use of combined right and left radial access: in this case, each ITA can be considered per se with the left ITA engaged from the left RA and the right ITA from the right RA. This approach could be considered in patients with high bleeding risk or significant contraindications to FA; the most important disadvantage for the patient is the combined cannulation of both radial arteries.
As far as venous aortocoronary grafts are concerned, there are no major anatomical constraints favoring left or right; nevertheless, when the catheter is coming from the left radial artery, similarly to the femoral, the operator has to deal with one area of resistance, whereas when the catheter is coming from the right radial artery, there are two areas of resistance affecting the torque of the catheter.14 Therefore, the engagement of venous grafts is usually easier with left than with right RA.
CONCLUSION
Although firm evidence from a large, adequately powered randomized controlled trial is lacking, RA offers several advantages compared to FA in performing diagnostic and interventional procedures in patients who have undergone previous CABG. First, RA is associated with a significant reduction in access-site complications and bleeding; this is clinically relevant because previous CABG was reported in approximately 18% of non–ST-acute coronary syndrome patients undergoing PCI,15 which, according to the European Society of Cardiology guidelines, should be performed via RA (class I recommendation, level of evidence A). Second, left RA is a straightforward approach to study the left ITA, which represents by far the most prevalent (and often the only) arterial graft in clinical practice. Nevertheless, RA presents several limitations, such as an increased radiation exposure and an increased need for crossover to another (usually femoral) access.
The appropriate selection of vascular access in CABG patients should be determined by both the operator’s experience and patient’s characteristics. Stable patients at low risk for femoral vascular complications would probably do better with FA in the hands of an operator who is at the beginning of his learning curve in RA. Conversely, an obese, anticoagulated patient with an occluded left radial artery and left ITA graft would probably do better with a right RA performed by a very skilled radial operator.
1. Ferrante G, Rao SV, Jüni P, et al. Radial versus femoral access for coronary interventions across the entire spectrum of patients with coronary artery disease: a meta-analysis of randomized trials. JACC Cardiovasc Interv. 2016;9:1419-1434.
2. Feldman DN, Swaminathan RV, Kaltenbach LA, et al. Adoption of radial access and comparison of outcomes to femoral access in percutaneous coronary intervention: an updated report from the national cardiovascular data registry (2007-2012). Circulation. 2013;127:2295-2306.
3. Aboyans V, Lacroix P, Postil A, et al. Subclinical peripheral arterial disease and incompressible ankle arteries are both long-term prognostic factors in patients undergoing coronary artery bypass grafting. J Am Coll Cardiol. 2005;46:815-820.
4. Michael TT, Alomar M, Papayannis A, et al. A randomized comparison of the transradial and transfemoral approaches for coronary artery bypass graft angiography and intervention: the RADIAL-CABG Trial (RADIAL Versus Femoral Access for Coronary Artery Bypass Graft Angiography and Intervention). JACC Cardiovasc Interv. 2013;6:1138-1144.
5. Sanmartin M, Cuevas D, Moxica J, et al. Transradial cardiac catheterization in patients with coronary bypass grafts: feasibility analysis and comparison with transfemoral approach. Catheter Cardiovasc Interv. 2006;67:580-584.
6. Rathore S, Roberts E, Hakeem AR, et al. The feasibility of percutaneous transradial coronary intervention for saphenous vein graft lesions and comparison with transfemoral route. J Interv Cardiol. 2009;22:336-340.
7. Bundhoo SS, Earp E, Ivanauskiene T, et al. Saphenous vein graft percutaneous coronary intervention via radial artery access: safe and effective with reduced hospital length of stay. Am Heart J. 2012;164:468-472.
8. Rigattieri S, Sciahbasi A, Brilakis ES, et al. Meta-analysis of radial versus femoral artery approach for coronary procedures in patients with previous coronary artery bypass grafting. Am J Cardiol. 2016;117:1248-1255.
9. Kedev S, Zafirovska B, Dharma S, Petkoska D. Safety and feasibility of transulnar catheterization when ipsilateral radial access is not available. Catheter Cardiovasc Interv. 2014;83:E51-E60.
10. Farooq V, Mamas MA, Fath-Ordoubadi F, et al. The use of a guide catheter extension system as an aid during transradial percutaneous coronary intervention of coronary artery bypass grafts. Catheter Cardiovasc Interv. 2011;78:847-863.
11. Patel T, Shah S, Pancholy S, et al. Balloon-assisted tracking: a must-know technique to overcome difficult anatomy during transradial approach. Catheter Cardiovasc Interv. 2014;83:211-220.
12. Locker C, Schaff HV, Dearani JA, et al. Multiple arterial grafts improve late survival of patients undergoing coronary artery bypass graft surgery: analysis of 8622 patients with multivessel disease. Circulation. 2012;126:1023-1030.
13. Cha KS, Kim MH. Feasibility and safety of concomitant left internal mammary arteriography at the setting of the right transradial coronary angiography. Catheter Cardiovasc Interv. 2002;56:188-195.
14. Patel T, Shah S, Pancholy S, et al. Native coronary and bypass graft cannulation through transradial approach: technical considerations. J Invasive Cardiol. 2015;27:E182-E189.
15. Nikolsky E, McLaurin BT, Cox DA, et al. Outcomes of patients with prior coronary artery bypass grafting and acute coronary syndromes: analysis from the ACUITY (Acute Catheterization and Urgent Intervention Triage Strategy) trial. JACC Cardiovasc Interv. 2012;5:919-926.
Stefano Rigattieri, MD, PhD
Interventional Cardiology Unit
Sandro Pertini Hospital
Rome, Italy
+39 06 41433870; stefanorigattieri@yahoo.it
Disclosures: None.
Alessandro Sciahbasi, MD, PhD
Interventional Cardiology Unit
Sandro Pertini Hospital
Rome, Italy
Disclosures: None.
Advertisement
Advertisement
