Advertisement
Advertisement
February 5, 2024
Procyrion Inc. Completes $57.7 Million Series E Funding Round
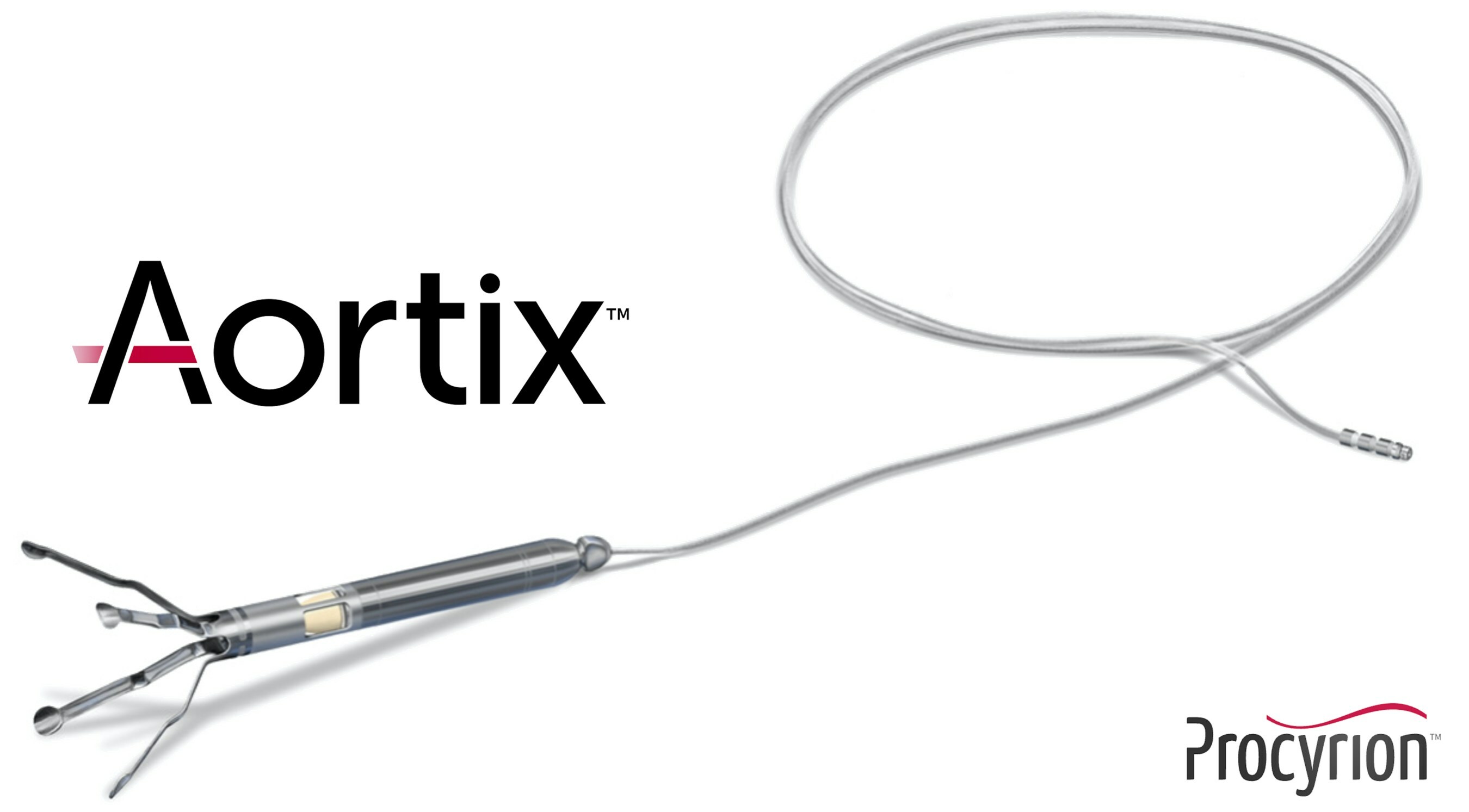 February 5, 2024—With funds from its latest Series E funding round, worth $57.7 million, Procyrion Inc. plans continued support for its DRAIN-HF pivotal investigational device exemption trial. DRAIN-HF is designed to evaluate the company’s Aortix percutaneous mechanical circulatory support (pMCS) device in patients with acute decompensated heart failure.
February 5, 2024—With funds from its latest Series E funding round, worth $57.7 million, Procyrion Inc. plans continued support for its DRAIN-HF pivotal investigational device exemption trial. DRAIN-HF is designed to evaluate the company’s Aortix percutaneous mechanical circulatory support (pMCS) device in patients with acute decompensated heart failure.
According to the company, Aortix pMCS is a catheter-deployed pump placed in the descending thoracic aorta and designed to directly increase perfusion to the kidneys while unloading the heart and improving cardiac performance. The device is designed to harness fluid entrainment to pump blood without the need for a valve, which may deliver a therapeutic benefit via a physiologically natural mechanism of action.
Last year, Procyrion reported initial findings from their Aortic CRS pilot study that showed patients demonstrated rapid decongestion with substantial removal of excess fluid and improved hemodynamics, as well as significant improvements in kidney function, cardiac function, and patient-reported assessment of shortness of breath sustained out to 30 days after treatment with the Aortix pump.
The round of funding was led by Fannin Partners in partnership with new and existing family/multifamily office investors as well as returning investors, including Bluebird Ventures and an undisclosed strategic investor.
In addition to the support of DRAIN-HF, the funds will also be used to support internal research and development programs to improve manufacturability and commercialization preparation for the company.
Advertisement
Advertisement
