Advertisement
Advertisement
January/February 2021
Physiologic Optimization of Revascularization
A review of post-PCI research and a discussion of improving patient prognosis and the benefits of physiologic assessments.
By Seung Hun Lee, MD, PhD; Doosup Shin, MD; Ki Hong Choi, MD; and Joo Myung Lee, MD, MPH, PhD
Invasive physiologic assessment with fractional flow reserve (FFR) or nonhyperemic pressure ratios (NHPRs), such as instantaneous wave-free ratio (iFR), is standard care for patients with coronary artery disease and is supported by the latest guidelines.1,2 However, FFR- or NHPR-guided percutaneous coronary intervention (PCI) do not necessarily yield “functionally complete or optimized revascularization,” even after angiographically successful PCI. Several studies have demonstrated that a substantial proportion of patients who underwent angiographically successful PCI had suboptimal post-PCI FFR3-5 or NHPR6-9 that was independently associated with worse clinical outcomes.3-8 Recent research has shown the clinical relevance of physiology-guided procedural optimization to achieve optimal post-PCI physiologic results and improve patient prognosis.10 In addition to the absolute values of post-PCI physiologic indices, multiple studies have shown physiologic gain (degree of change in FFR after PCI) to be significantly associated with improvements in angina severity, quality of life, and future prognosis after revascularization.11-13 Nevertheless, there is no standard recommendation or consensus on the proper use of FFR or NHPRs after PCI. This article discusses the integration of post-PCI physiologic assessment in daily practice and optimization of procedural results to improve patient prognosis.
POST-PCI PHYSIOLOGIC Indices AS PROGNOSTIC INDICATORS
Over the last decade, ample evidence has shown that post-PCI FFR conveys valuable information regarding the functional results of revascularization, with prognostic implications.10-27 Although optimal cut-off values for post-PCI FFR vary between 0.86 and 0.96, most studies, including meta-analyses, have reported similar inverse relationships between post-PCI FFR values and risk of future clinical events. It should be noted that the designs of studies defining optimal cut-off values for post-PCI FFR were different from those of pre-PCI FFR studies. The cut-off values of pre-PCI FFR were validated using three noninvasive stress tests to detect myocardial ischemia. Conversely, the cut-off values of post-PCI FFR were evaluated to predict the occurrence of clinical events. Therefore, these values may vary according to the study population, event rates, or definition of primary events. Nevertheless, post-PCI FFR is independently predictive of long-term clinical outcomes, and the inverse relationship between post-PCI FFR and risk of clinical events was consistently observed across studies.
Recent studies have evaluated the prognostic implications of post-PCI NHPRs.6-9 Hakeem et al evaluated 574 patients and reported that compared with satisfactory post-PCI distal-to-aortic pressure ratio (Pd/Pa) (> 0.96), those with suboptimal post-PCI Pd/Pa (≤ 0.96) had a significantly higher risk of major adverse cardiovascular events (MACEs), which was a composite of death, myocardial infarction (MI), and target vessel revascularization (TVR) (15% vs 24%; log-rank P = .0006).7 The DEFINE-PCI study was a single-arm registry that evaluated post-PCI iFR after angiographically successful PCI among 500 patients.6,9 In that study, 21.9% of vessels (24% of patients) had an ischemic range of post-PCI iFR ≤ 0.89 despite angiographically successful PCI. Based on post-PCI iFR pullback, 81.6% of patients with post-PCI iFR ≤ 0.89 had focal stenosis that was angiographically unapparent and 18.4% had diffuse disease. The optimal cut-off value of post-PCI iFR for cardiac death or spontaneous MI after 1 year was 0.95 (< 0.95 vs ≥ 0.95; 3.2% vs 0%; log-rank P = .02).9 A multicenter study using the PERSPECTIVE-PCI registry evaluated 588 patients who underwent PCI with both post-PCI resting Pd/Pa and post-PCI FFR measurements. That study showed significant difference in target vessel failure (TVF; a composite of cardiac death, target vessel–related MI, and clinically driven TVR) according to ischemic cut-off values for post-PCI FFR (≤ 0.80 vs > 0.80; 10.3% vs 2.5%; P < .001) and Pd/Pa (≤ 0.92 vs > 0.92; 6.2% vs 2.5%; P = .029).8
PHYSIOLOGY-GUIDED PROCEDURAL OPTIMIZATION
The results and consistent observations of discordance between angiographic and functional results of PCI support the potential role of post-PCI physiologic assessment as a functional optimization tool and gatekeeper to decide further interventional procedures and optimize final results. Evaluation of the potential mechanism of suboptimal post-PCI physiologic results should be performed before determining whether additional interventional procedures are required.
The FFR-SEARCH study evaluated the potential mechanisms of suboptimal post-PCI FFR using high-definition intravascular ultrasound (IVUS).28 In 100 vessels with post-PCI FFR ≤ 0.85, IVUS identified stent underexpansion (< 90% of average reference lumen area) in 74% of vessels; a significant residual focal lesion in proximal or distal segments in 29% and 30%, respectively; stent malapposition in 23% (87% were accompanied by underexpansion); vascular spasm in 9%; residual diffuse atherosclerotic disease in 8%; and lumen-compromising intramural hematoma in 3%. Similarly, Wolfrum et al reported optical coherence tomography (OCT) findings for 21 patients with post-PCI FFR < 0.90.5 Among these patients, 13 (61.9%) showed OCT-defined suboptimal stent deployment and underwent additional interventional procedures. The primary reasons for OCT-defined suboptimal stent deployment were stent underexpansion (46%), incomplete lesion coverage (39%), stent malapposition (54%; all cases of malapposition were accompanied by stent underexpansion and/or incomplete lesion coverage), stent distal edge dissection (15%), and tissue protrusion (8%). In eight (38.1%) patients with post-PCI FFR < 0.90, there was no significant issue in the stented segment, but diffuse atherosclerotic disease was found at the distal segment of the vessel. After OCT-guided stent optimization, post-PCI FFR increased significantly from 0.80 ± 0.02 to 0.88 ± 0.01 (P = .008).
The concept of post-PCI FFR-guided functional optimization with additional procedures was presented by Agarwal et al.10 Among 574 patients, 21% of lesions showed persistent ischemic post-PCI FFR ≤ 0.80 after angiographically successful PCI. Among these lesions, operators performed adjunctive postdilatation in 42%, additional stenting in 33%, combined adjunctive postdilatation and stenting in 18%, and intravascular imaging evaluation in 9%. These additional interventions significantly reduced the proportion of lesions with ischemic FFR from 21% to 9%. A recent prospective registry of 230 lesions (206 patients) evaluated the clinical relevance of functional optimization of PCI using post-PCI FFR where 36.5% of revascularized vessels showed immediate post-PCI FFR ≤ 0.80.29 Among these vessels, further intervention was required based on post-PCI FFR pullback tracing, and post-PCI FFR significantly increased from 0.73 (Q1–Q3: 0.69–0.77) to 0.80 (Q1–Q3: 0.77–0.85) after subsequent intervention (P < .0001). In a recent randomized controlled trial (TARGET-FFR), patients who underwent angiographically successful PCI were randomized to a physiology-guided incremental optimization strategy (PIOS) versus a blinded control group. In the PIOS group, adjunctive balloon dilation or additional stenting was performed if there was a focal FFR increase ≥ 0.05 in the stented or nonstented segment, respectively. Although PIOS failed to improve the proportion of patients with optimal post-PCI FFR ≥ 0.90 (38.1% vs 28.1%; P = .099), it was associated with a significantly smaller proportion of patients with post-PCI FFR ≤ 0.80 compared with the control group (18.6% vs 29.8%; P = .045).30,31
Figure 1 shows practical case examples involving post-PCI physiology-guided procedural optimization. In both cases, immediate post-PCI FFR values were in the ischemic range, and pullback pressure tracings revealed focal step-up (orange arrows). Intravascular imaging or careful angiographic assessment showed stent underexpansion (Figure 1A) or distal edge dissection (Figure 1B). After additional procedures such as adjunctive balloon dilatation or additional stenting, final post-PCI FFR values were improved.
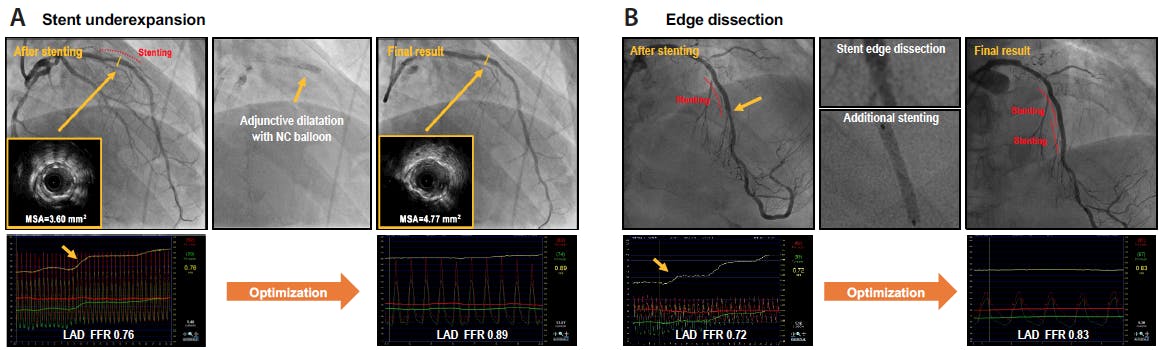
Figure 1. Physiology-guided procedural optimization. Illustrative case examples showing the benefit of post-PCI physiologic assessment on the optimization of the final results. After stenting at mid-left anterior descending (LAD) artery, immediate post-PCI FFR was 0.76, with focal step-up at the stented segment. IVUS revealed stent underexpansion, which was corrected by high-pressure adjunctive balloon inflation with a noncompliant (NC) balloon. After procedural optimization, the final post-PCI FFR increased to 0.89 (A). After stenting at mid-LAD, immediate post-PCI FFR was 0.72, with significant focal step-up at the distal edge of the stent. Careful examination revealed distal edge dissection. Additional stent implantation improved the final post-PCI FFR to 0.83 (B). MSA, minimum stent area.
PHYSIOLOGIC GAIN AFTER PCI AND DISEASE PATTERNS
In addition to the absolute value of post-PCI physiologic indices and physiology-guided procedural optimization, physiologic gain from stenting should be considered. Physiologic gain is defined as absolute or relative changes in physiologic index after PCI (∆FFR = post-PCI FFR – pre-PCI FFR; percent FFR increase = [post-PCI FFR – pre-PCI FFR]/pre-PCI FFR x 100). PCI with stent implantation is a local treatment, and post-PCI FFR reflects both residual stenosis in the stented segment and residual disease beyond the stented segment in the target vessel. Therefore, a single cut-off value for post-PCI FFR cannot fully discriminate between the relative contributions of stented and nonstented disease burden to patient prognosis. In this regard, quantification of FFR gains from PCI enables us to evaluate the contribution of local stenosis treated by PCI to the total cumulative disease burden in the target vessel. Previous studies have evaluated the concept of FFR gains from PCI and their clinical relevance.11-13 In these studies, greater increase in ∆FFR or percent FFR increase were significantly associated with improved quality of life, angina class, reduced vessel-oriented composite endpoint, and TVF at 2 years.11-13
The patterns of FFR gains after PCI and absolute values of post-PCI FFR are determined mainly by underlying patterns of coronary atherosclerotic disease or plaque distribution in the target vessels (Figure 2). In predominant focal disease, PCI with stenting offers greater physiologic gain, satisfactory post-PCI physiologic results, and favorable clinical outcomes. Conversely, in predominant diffuse disease, the effect of PCI on reducing target vessel ischemic burden is limited, resulting in less physiologic gain, suboptimal post-PCI physiologic results, and worse clinical outcomes. Therefore, if both post-PCI FFR and physiologic gain are suboptimal despite procedural optimization, the benefit of additional local treatment would be limited; thus, meticulous secondary prevention and close follow-up are warranted.
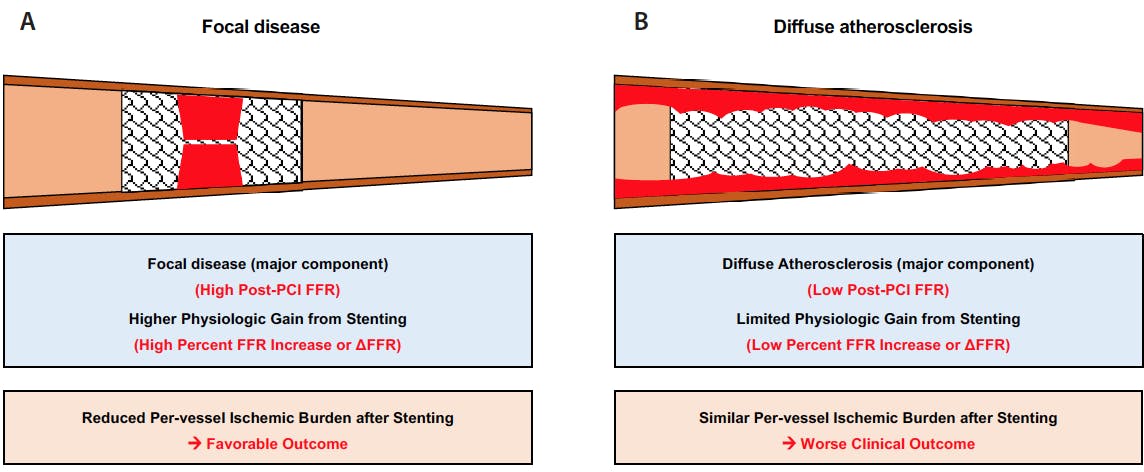
Figure 2. Physiologic response to focal treatment depends on disease characteristics. Because physiologic indexes such as FFR or NHPRs are per-vessel indexes, whereas PCI with stenting is a focal treatment, the physiologic response of stenting varies by disease characterization: focal disease (A) versus diffuse atherosclerosis (B). Therefore, additional consideration of physiologic gain from stenting should be considered in addition to the absolute post-PCI values of physiologic indexes.
PROGNOSTIC IMPACT OF FUNCTIONALLY OPTIMIZED REVASCULARIZATION
Previous studies showed that residual angiographic disease, which was assessed by residual SYNTAX score (RSS), possessed prognostic impact after angiographically successful PCI.32,33 However, given the significantly reduced number of stents with improved clinical outcome after FFR-guided PCI,34 it is unknown whether residual angiographic disease after FFR-guided PCI yields a similar prognostic impact. Kobayashi et al published a post hoc analysis of the FAME trial that showed no significant difference in RSS between patients with and without MACE at 1 year after FFR-guided revascularization for multivessel disease.35 A later patient-level pooled analysis of the DANAMI-3-PRIMULTI, FAME, and FAMOUS-NSTEMI trials showed consistent results for patients with acute coronary syndrome.36 In these studies, FFR-guided PCI meant that FFR was used to perform (FFR ≤ 0.80) or defer (FFR > 0.80) revascularization. Considering that approximately 20% of patients showed post-PCI FFR ≤ 0.80 even after angiographically successful PCI,10,11 the prognostic impact of residual angiographic disease should be evaluated after functionally optimized revascularization without residual myocardial ischemia, defined as post-PCI FFR > 0.80.
A recent study from the International Post PCI FFR Registry evaluated 1,910 patients with 2,095 revascularized vessels with post-PCI FFR > 0.80.37 TVF occurred after 2 years in 4.9% of the study population, and patients with TVF had higher pre-PCI SYNTAX score and lower post-PCI FFR than those without. However, there was no significant difference in RSS between the two groups. Furthermore, risk of TVF was significantly different according to tertile of post-PCI FFR (log-rank P = .009) but not RSS (log-rank P = .851). These differences in prognostic impact between post-PCI FFR and RSS suggest that post-PCI FFR is a better tool to assess residual coronary atherosclerotic disease burden than is angiographic assessment, supporting the importance of functionally optimized revascularization.
ADDITIONAL CONSIDERATIONS FOR POST-PCI PHYSIOLOGIC ASSESSMENT
Applying post-PCI physiologic assessment in daily practice requires an understanding of its potential limitations. Any invasive physiologic indices can be influenced by transient changes in boundary conditions, such as driving pressure, microcirculatory resistance, or coronary flow. Microcirculatory resistance is the main determinant of coronary flow through an epicardial artery, and procedure-related transient microcirculatory dysfunction can influence post-PCI FFR value. Murai et al compared post-PCI FFR values according to the index of microcirculatory resistance (IMR) measured after successful elective stenting.38 A total of 104 patients were classified according to post-PCI IMR quartile. Although there was no significant difference in angiographic residual stenosis among quartiles, there was a significant linear association between post-PCI IMR and FFR values. These results indicate that prognostic stratification by post-PCI FFR or FFR gain from PCI can be limited in the presence of combined severe microvascular disease or transient microvascular dysfunction after PCI. Therefore, caution is needed when interpreting post-PCI FFR if significant microvascular dysfunction, such as no reflow or slow flow, occurred during the procedure.
Post-PCI NHPRs can also be influenced by transient changes in boundary conditions such as increased heart rate, elevated sympathetic tone, or postocclusion reactive hyperemia. In the PERSPECTIVE-PCI registry, 93.8% of patients with post-PCI Pd/Pa > 0.92 had concordant post-PCI FFR (> 0.80), yet 60.4% of patients with post-PCI Pd/Pa ≤ 0.92 had discordant post-PCI FFR (> 0.80) (Figure 3, lower left panel).8 Regarding clinical outcomes, only patients with concordantly suboptimal post-PCI Pd/Pa ≤ 0.92 and FFR ≤ 0.80 had increased risk of TVF. In contrast, patients with suboptimal post-PCI Pd/Pa ≤ 0.92 but negative FFR > 0.80 had a similar risk for TVF to patients with post-PCI Pd/Pa > 0.92 (Figure 3, lower right panel). Furthermore, when comparing resting and hyperemic mean transit time (Tmn), patients with post-PCI Pd/Pa ≤ 0.92 but FFR > 0.80 had significantly lower resting Tmn than those with post-PCI Pd/Pa > 0.92. However, there was no significant difference in hyperemic Tmn between the two groups. These results suggest that suboptimal post-PCI Pd/Pa values might not be caused entirely by residual ischemia, but also by increased resting coronary flow in the post-PCI phase. Previous studies consistently have demonstrated the presence of postocclusion reactive hyperemia,39-41 which was observed to occur even after brief coronary occlusion (< 2 seconds in duration).42 Therefore, post-PCI NHPRs can be influenced by postocclusion reactive hyperemia and can result in a high rate of discordance between post-PCI NHPRs and FFR. Because there is no available index or method to confirm that patients have recovered to baseline or resting status in the post-PCI phase, reassessment of post-PCI FFR is warranted in patients whose ischemic range of post-PCI NHPRs has no reasonable explanation based on angiographic or intravascular imaging findings. If NHPRs are the only available option in the cardiac catheterization laboratory, the operator should be cautious to ensure sufficient recovery time before measuring post-PCI NHPRs.
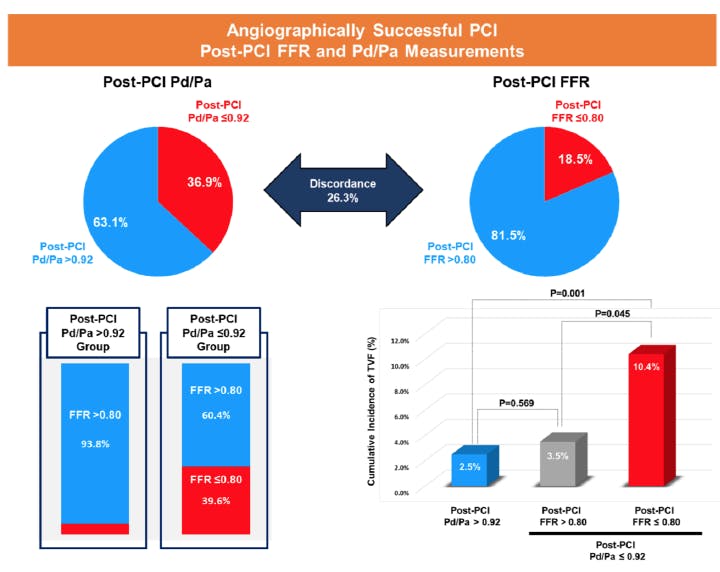
Figure 3. Prognostic implications of post-PCI hyperemic and nonhyperemic indexes. Post-PCI NHPRs can be influenced by transient changes in boundary conditions such as increased heart rate, elevated sympathetic tone, or postocclusion reactive hyperemia and can be abnormal. A recent study showed that the majority of abnormal post-PCI NHPRs had sufficient post-PCI FFR, and the positive predictive value of post-PCI NHPRs for adverse clinical outcomes was relatively lower than that of post-PCI FFR. Reprinted with permission. This article was published in JACC Cardiovascular Interventions, Vol. 13, Shin D, Lee SH, Lee JM, et al. Prognostic implications of post-intervention resting Pd/Pa and fractional flow reserve in patients with stent implantation, 1920-1933, Copyright Elsevier (2020).
PRACTICAL POINTS FOR POST-PCI PHYSIOLOGIC ASSESSMENT
These results support post-PCI FFR as a reliable indicator of suboptimal stent deployment and a signal for the need of a subsequent interventional procedure. Because most patients with negative post-PCI NHPRs also had negative post-PCI FFR, post-PCI NHPRs could be measured first. If post-PCI NHPR is negative, induction of hyperemia to measure FFR is not necessary. If post-PCI NHPR is positive, confirmation by post-PCI FFR is warranted. When post-PCI FFR is lower than expected, it is important to identify a correctable mechanism. Pressure pullback tracings would be helpful for discriminating focal step-up across the stented and nonstented segments from diffuse gradual step-up at the distal segment of the vessel. In the presence of focal step-up, intravascular imaging could reveal the target of a subsequent interventional procedure. If the final post-PCI FFR is suboptimal even after a successful additional procedure, repeated pressure pullback tracings would be helpful to discriminate residual focal stenosis from diffuse gradual step-up at the distal segment of the vessel. Further stent implantation would have limited benefit for diffuse gradual step-up compared with recommended medical treatments. In such cases, FFR gain from PCI can provide information about physiologic gain from stent implantation and whether any possible benefit can be gained from additional local treatment (Figure 4).11
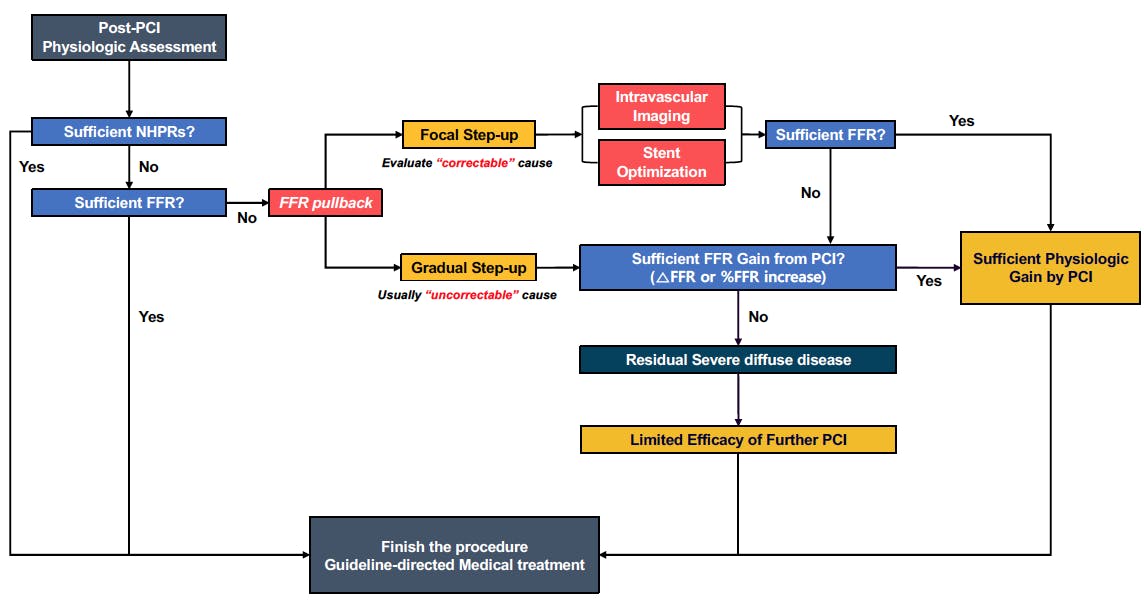
Figure 4. Suggested algorithm of post-PCI physiologic assessment. After revascularization, post-PCI physiologic assessment should be performed to functionally optimize the results. In satisfactory post-PCI NHPRs, the procedure can be completed without additional evaluation. However, if the post-PCI NHPRs are insufficient, FFR measurement is recommended, and FFR pullback curve analysis should be performed to evaluate the cause of the suboptimal physiologic results. Regarding the step-up patterns at the pullback curve, intravascular imaging is required to determine a further treatment plan. An insufficient final physiologic result with a gradual step-up pattern indicates residual diffuse atherosclerotic disease, in which additional procedures would not improve the final physiologic results.
CONCLUSION
The purpose of revascularization is not only to alleviate angiographic stenosis, but also to resolve myocardial ischemia and enhance patient prognosis. Post-PCI physiologic assessment with FFR or NHPRs provides information about the functional results of revascularization and prognostic stratification after PCI. In cases with suboptimal post-PCI physiologic results, further investigations and interventions aimed to find potential causes can improve final results and patient prognosis. The absolute values of post-PCI physiologic indices and physiologic gain from PCI are important to further stratify patients with higher risk of future clinical events. Physiologic assessments by FFR or NHPRs are practical and applicable tools enabling not just the determination of revascularization but also functional optimization of PCI to improve outcomes.
1. Neumann FJ, Sousa-Uva M, Ahlsson A, et al. 2018 ESC/EACTS Guidelines on myocardial revascularization. Eur Heart J. 2019;40:87-165. doi: 10.1093/eurheartj/ehy394
2. Patel MR, Calhoon JH, Dehmer GJ, et al. ACC/AATS/AHA/ASE/ASNC/SCAI/SCCT/STS 2017 appropriate use criteria for coronary revascularization in patients with stable ischemic heart disease: a report of the American College of Cardiology Appropriate Use Criteria Task Force, American Association for Thoracic Surgery, American Heart Association, American Society of Echocardiography, American Society of Nuclear Cardiology, Society for Cardiovascular Angiography and Interventions, Society of Cardiovascular Computed Tomography, and Society of Thoracic Surgeons. J Am Coll Cardiol. 2017;69:2212-2241. doi: 10.1016/j.jacc.2017.02.001
3. Johnson NP, Tóth GG, Lai D, et al. Prognostic value of fractional flow reserve: linking physiologic severity to clinical outcomes. J Am Coll Cardiol. 2014;64:1641-1654. doi: 10.1016/j.jacc.2014.07.973
4. Rimac G, Fearon WF, De Bruyne B, et al. Clinical value of post-percutaneous coronary intervention fractional flow reserve value: a systematic review and meta-analysis. Am Heart J. 2017;183:1-9. doi: 10.1016/j.ahj.2016.10.005
5. Wolfrum M, Fahrni G, de Maria GL, et al. Impact of impaired fractional flow reserve after coronary interventions on outcomes: a systematic review and meta-analysis. BMC Cardiovasc Disord. 2016;16:177. doi: 10.1186/s12872-016-0355-7
6. Jeremias A, Davies JE, Maehara A, et al. Blinded physiological assessment of residual ischemia after successful angiographic percutaneous coronary intervention: the DEFINE PCI Study. JACC Cardiovasc Interv. 2019;12:1991-2001. doi: 10.1016/j.jcin.2019.05.054
7. Hakeem A, Ghosh B, Shah K, et al. Incremental prognostic value of post-intervention Pd/Pa in patients undergoing ischemia-driven percutaneous coronary intervention. JACC Cardiovasc Interv. 2019;12:2002-2014. doi: 10.1016/j.jcin.2019.07.026
8. Shin D, Lee SH, Lee JM, et al. Prognostic implications of post-intervention resting Pd/Pa and fractional flow reserve in patients with stent implantation. JACC Cardiovasc Interv. 2020;13:1920-1933. doi: 10.1016/j.jcin.2020.05.042
9. Patel M, Jeremias A, Davies J, et al. 1-year outcomes of patients with residual physiologic ischemia after percutaneous coronary intervention: The DEFINE PCI Trial. Presented at: TCT 2020. October 15, 2020.
10. Agarwal SK, Kasula S, Hacioglu Y, et al. Utilizing post-intervention fractional flow reserve to optimize acute results and the relationship to long-term outcomes. JACC Cardiovasc Interv. 2016;9:1022-1031. doi: 10.1016/j.jcin.2016.01.046
11. Lee JM, Hwang D, Choi KH, et al. Prognostic implications of relative increase and final fractional flow reserve in patients with stent implantation. JACC Cardiovasc Interv. 2018;11:2099-2109. doi: 10.1016/j.jcin.2018.07.031
12. Fournier S, Ciccarelli G, Toth GG, et al. Association of improvement in fractional flow reserve with outcomes, including symptomatic relief, after percutaneous coronary intervention. JAMA Cardiol. 2019;4:370-374. doi: 10.1001/jamacardio.2019.0175
13. Nishi T, Piroth Z, De Bruyne B, et al. Fractional flow reserve and quality-of-life improvement after percutaneous coronary intervention in patients with stable coronary artery disease. Circulation. 2018;138:1797-1804. doi: 10.1161/CIRCULATIONAHA.118.035263
14. Pijls NH, Klauss V, Siebert U, et al. Coronary pressure measurement after stenting predicts adverse events at follow-up: a multicenter registry. Circulation. 2002;105:2950-2954. doi: 10.1161/01.cir.0000020547.92091.76
15. Dupouy P, Gilard M, Morelle JF, et al. Usefulness and clinical impact of a fractional flow reserve and angiographic targeted strategy for coronary artery stenting: FROST III, a multicenter prospective registry. EuroIntervention. 2005;1:85-92.
16. Klauss V, Erdin P, Rieber J, et al. Fractional flow reserve for the prediction of cardiac events after coronary stent implantation: results of a multivariate analysis. Heart. 2005;91:203-206. doi: 10.1136/hrt.2003.027797
17. Jensen LO, Thayssen P, Thuesen L, et al. Influence of a pressure gradient distal to implanted bare-metal stent on in-stent restenosis after percutaneous coronary intervention. Circulation. 2007;116:2802-2808. doi: 10.1161/CIRCULATIONAHA.107.704064
18. Ishii H, Kataoka T, Kobayashi Y, et al. Utility of myocardial fractional flow reserve for prediction of restenosis following sirolimus-eluting stent implantation. Heart Vessels. 2011;26:572-581. doi: 10.1007/s00380-010-0105-1
19. Leesar MA, Satran A, Yalamanchili V, et al. The impact of fractional flow reserve measurement on clinical outcomes after transradial coronary stenting. EuroIntervention. 2011;7:917-923. doi: 10.4244/EIJV7I8A145
20. Nam CW, Hur SH, Cho YK, et al. Relation of fractional flow reserve after drug-eluting stent implantation to one-year outcomes. Am J Cardiol. 2011;107:1763-1767. doi: 10.1016/j.amjcard.2011.02.329
21. Ito T, Tani T, Fujita H, Ohte N. Relationship between fractional flow reserve and residual plaque volume and clinical outcomes after optimal drug-eluting stent implantation: insight from intravascular ultrasound volumetric analysis. Int J Cardiol. 2014;176:399-404. doi: 10.1016/j.ijcard.2014.07.115
22. Doh JH, Nam CW, Koo BK, et al. Clinical relevance of poststent fractional flow reserve after drug-eluting stent implantation. J Invasive Cardiol. 2015;27:346-351.
23. Reith S, Battermann S, Hellmich M, et al. Correlation between OCT-derived intrastent dimensions and fractional flow reserve measurements after coronary stent implantation and impact on clinical outcome. J Invasive Cardiol. 2015;27:222-228.
24. Kasula S, Agarwal SK, Hacioglu Y, et al. Clinical and prognostic value of poststenting fractional flow reserve in acute coronary syndromes. Heart. 2016;102:1988-1994. doi: 10.1136/heartjnl-2016-309422
25. Li SJ, Ge Z, Kan J, et al. Cutoff value and long-term prediction of clinical events by FFR measured immediately after implantation of a drug-eluting stent in patients with coronary artery disease: 1- to 3-year results from the DKCRUSH VII Registry Study. JACC Cardiovasc Interv. 2017;10:986-995. doi: 10.1016/j.jcin.2017.02.012
26. Piroth Z, Toth GG, Tonino PAL, et al. Prognostic value of fractional flow reserve measured immediately after drug-eluting stent implantation. Circ Cardiovasc Interv. 2017;10:e005233. doi: 10.1161/CIRCINTERVENTIONS.116.005233
27. Hwang D, Lee JM, Lee HJ, et al. Influence of target vessel on prognostic relevance of fractional flow reserve after coronary stenting. EuroIntervention. 2019;15:457-464. doi: 10.4244/EIJ-D-18-00913
28. van Zandvoort LJC, Masdjedi K, Witberg K, et al. Explanation of postprocedural fractional flow reserve below 0.85. Circ Cardiovasc Interv. 2019;12:e007030. doi: 10.1161/CIRCINTERVENTIONS.118.007030
29. Uretsky BF, Agarwal SK, Vallurupalli S, et al. Prospective evaluation of the strategy of functionally optimized coronary intervention. J Am Heart Assoc. 2020;9:e015073. doi: 10.1161/JAHA.119.015073
30. Collison D, McClure JD, Berry C, Oldroyd KG. A randomized controlled trial of a physiology-guided percutaneous coronary intervention optimization strategy: rationale and design of the TARGET FFR study. Clin Cardiol. 2020;43:414-422. doi: 10.1002/clc.23342
31. Collison D, McClure JD, Berry C, Oldroyd K. TARGET FFR - Physiology-guided optimization of PCI: a randomized controlled trial. Presented at: TCT 2020. October 16, 2020.
32. Farooq V, Serruys PW, Bourantas CV, et al. Quantification of incomplete revascularization and its association with five-year mortality in the synergy between percutaneous coronary intervention with taxus and cardiac surgery (SYNTAX) trial validation of the residual SYNTAX score. Circulation. 2013;128:141-151. doi: 10.1161/CIRCULATIONAHA.113.001803
33. Généreux P, Palmerini T, Caixeta A, et al. Quantification and impact of untreated coronary artery disease after percutaneous coronary intervention: the residual SYNTAX (synergy between PCI with Taxus and cardiac surgery) score. J Am Coll Cardiol. 2012;59:2165-2174. doi: 10.1016/j.jacc.2012.03.010
34. Xaplanteris P, Fournier S, Pijls NHJ, et al. Five-year outcomes with PCI guided by fractional flow reserve. N Engl J Med. 2018;379:250-259. doi: 10.1056/NEJMoa1803538
35. Kobayashi Y, Nam CW, Tonino PA, et al. The prognostic value of residual coronary stenoses after functionally complete revascularization. J Am Coll Cardiol. 2016;67:1701-1711. doi: 10.1016/j.jacc.2016.01.056
36. Kobayashi Y, Lønborg J, Jong A, et al. Prognostic value of the residual SYNTAX score after functionally complete revascularization in ACS. J Am Coll Cardiol. 2018;72:1321-1329. doi: 10.1016/j.jacc.2018.06.069
37. Lee JM, Hwang D, Choi KH, et al. Prognostic impact of residual anatomic disease burden after functionally complete revascularization. Circ Cardiovasc Interv. 2020;13:e009232. doi: 10.1161/CIRCINTERVENTIONS.120.009232
38. Murai T, Lee T, Yonetsu T, et al. Influence of microvascular resistance on fractional flow reserve after successful percutaneous coronary intervention. Catheter Cardiovasc Interv. 2015;85:585-592. doi: 10.1002/ccd.25499
39. Kawase Y, Omori H, Kawasaki M, et al. Postocclusional hyperemia for fractional flow reserve after percutaneous coronary intervention. Circ Cardiovasc Interv. 2017;10:e005674. doi: 10.1161/CIRCINTERVENTIONS.117.005674
40. Serruys PW, Juillière Y, Zijlstra F, et al. Coronary blood flow velocity during percutaneous transluminal coronary angioplasty as a guide for assessment of the functional result. Am J Cardiol. 1988;61:253-259. doi: 10.1016/0002-9149(88)90926-5
41. Coffman JD, Gregg DE. Reactive hyperemia characteristics of the myocardium. Am J Physiol. 1960;199:1143-1149. doi: 10.1152/ajplegacy.1960.199.6.1143
42. Marcus M, Wright C, Doty D, et al. Measurements of coronary velocity and reactive hyperemia in the coronary circulation of humans. Circ Res. 1981;49:877-891. doi: 10.1161/01.res.49.4.877
Advertisement
Advertisement
