Advertisement
Advertisement
March/April 2018
Acute Right Ventricular Failure
A review of diagnosis and principles of percutaneous mechanical circulatory support to optimize RV preload, afterload, and contractility after acute RV failure.
Cardiogenic shock is defined as systemic tissue hypoperfusion secondary to inadequate cardiac output despite adequate circulatory volume and ventricular filling pressure. Diagnostic criteria for cardiogenic shock include a systolic blood pressure (BP) < 90 mm Hg for > 30 minutes; a decrease in mean arterial BP > 30 mm Hg below baseline, with a cardiac index < 1.8 L/min/m2 without hemodynamic support or < 2.2 L/min/m2 with support; and elevated ventricular filling pressure.1
Shock due to predominant acute right ventricular (RV) failure is characterized by clinical findings of hypotension; low cardiac output; compensatory increase in systemic vascular resistance often manifesting as cool, clammy extremities; clear lungs signifying normal left ventricular (LV) filling pressure; and elevated jugular venous pressure indicating increased RV preload. In acute RV dysfunction, the RV free wall (RVFW) is unable to contribute to stroke work, resulting in failure to maintain forward flow into the pulmonary artery (PA) and subsequent reduced LV preload. RV dilation shifts the interventricular septum toward the left ventricle, which further impairs LV preload, an effect further exacerbated by elevated intrapericardial pressure (a result of abrupt RV dilation). Under conditions of acute ischemic RVFW ischemic dysfunction, RV systolic pressure and global performance are generated by LV septal contractile contributions mediated via the interventricular septum.2-4 This cascade of events, if not promptly recognized and treated, can result in shock and death.
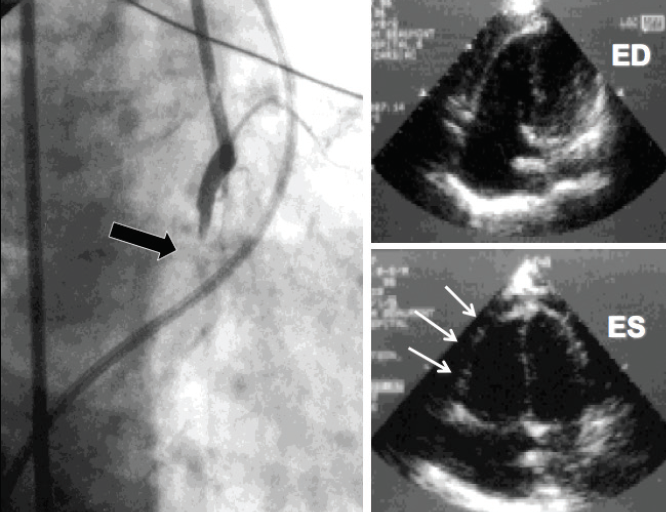
Figure 1. Acute IMI with RVI. Echocardiography reveals RV enlargement with dyskinetic free wall motion and septal bowing toward the relatively underfilled LV. Angiography reveals occlusion of proximal right coronary artery. ED, end diastolic; ES, end systole. Adapted from Bowers TR, O’Neill WO, Pica M, Goldstein JA. Patterns of coronary compromise resulting in acute right ventricular ischemic dysfunction. Circulation. 2002;106:1104–1109.
Acute RV failure may be associated with acute inferior myocardial infarction (IMI) with RV involvement (RVI) (Figure 1), post–LV assist device (LVAD) implantation, postorthotopic heart transplantation, postcardiotomy after any cardiac surgical procedure, acute pulmonary embolism, and acute right heart valve insufficiency (eg, acute tricuspid or pulmonic valve regurgitation). Echocardiography provides evidence of RV failure, including RV dilation, depressed RVFW motion, and diminished RV ejection fraction. The right ventricle may be larger than the left ventricle (RV/LV end-diastolic area ratio > 0.6), and reverse septal curvature is often present.5 Echocardiography is also useful to exclude pericardial disease, which is beyond the scope of this review.
The use of a PA catheter is indispensable in the contemporary management of cardiogenic shock,6 and shock due to acute RV failure is no exception (although caution must be employed when inserting any catheter into the ischemic dilated RV, which is very sensitive and prone to ventricular arrhythmias). Cardiac power output (CPO; expressed in Watts) is the single most predictive marker of prognosis in cardiogenic shock.7 It is easily calculated by multiplying mean systemic BP by cardiac output and dividing by a constant, 451 (CPO = [MAP X CO]/451). The ratio of central venous pressure to pulmonary capillary wedge pressure > 0.63 predicts RV failure after LVAD implantation.8 PA pulsatility index (PAPi) may be the most accurate predictor of RV failure after LVAD implantation or after IMI with RVI9,10 and can easily be calculated using commonly collected hemodynamic parameters (PAPi = [systolic PA pressure – diastolic PA pressure]/mean right atrial pressure).
MANAGEMENT
Medical Therapy
Medical therapy for acute RV failure is based on fundamental principles of optimizing preload, afterload, and contractility.11-13 Achieving acceptable RV preload can be accomplished with intravenous fluid resuscitation or diuresis/dialysis as appropriate. In most cases, maintaining central venous pressure around 8 to 12 mm Hg is optimal. If the RV is failing due to excessive afterload, this can be reduced using pulmonary vasodilators (or via thrombolysis in the case of thromboemboli). Vasopressors and inotropes may also be required to maintain an aortic BP sufficient to perfuse the coronary artery circulation and augment RV contractility. If these maneuvers fail to restore adequate tissue perfusion, mechanical circulatory support (MCS) may be required.
Revascularization
In the case of acute RV failure due to RVI, reperfusion of the occluded right coronary artery results in rapid restoration of RV systolic function and improved hemodynamics. In the SHOCK trial registry, patients with shock due to predominant RV failure had a similar mortality rate as patients with predominant LV failure (~50%).14 Achieving successful myocardial reperfusion appears to be a critical determinant of outcome. In a single-center series of 53 patients with acute RVI, achievement of angiographically normal flow in the right coronary artery and all its major branches resulted in dramatic recovery of RV performance, improved cardiac output, and improved survival (98% vs 42% with unsuccessful reperfusion; P = .001).15 Importantly, vagal-mediated bradycardia and/or heart block can be encountered prior to, during, and after revascularization, and temporary pacing may be required.
Thrombolytic therapy for acute massive or submassive pulmonary embolism results in restoration of pulmonary blood flow, reversal of compensatory pulmonary vasoconstriction in lung segments not directly affected by clot burden, reduction in RV afterload, increase in LV preload, and increase in systemic BP and cardiac output.16,17
It is essential to emphasize that the right ventricle has a remarkable ability to recover function in the setting of acute RVI with successful reperfusion (even after prolonged ischemia) or after relief of acute pressure overload following successful lysis of pulmonary thromboemboli.
Intra-Aortic Balloon Pump
Use of an intra-aortic balloon pump (IABP) is known to improve cardiac hemodynamics by increasing coronary artery perfusion and reducing LV afterload. Its effect on RV function in cardiogenic shock is not as well characterized. In a series of 32 patients with shock due to acute IMI and RVI treated with IABP and revascularization of culprit right coronary artery occlusion, survival was 81%.18 IABP augmented mean systemic BP in all patients, imparting benefits to systemic perfusion in general and specifically to right coronary artery perfusion during and after revascularization, as well as to the left coronary system, a critical benefit in the setting of shock with multivessel coronary artery disease. In addition, because the failing right ventricle depends on the interventricular septum to maintain stroke volume, reducing LV afterload with IABP may indirectly improve RV performance. This may be especially important in patients with both RVI and concomitant LV dysfunction, which is associated with worse outcomes than in RVI with intact LV function.19
Direct Right Ventricular Support
Commercially available devices that directly unload the right ventricle include Impella RP (Abiomed, Inc.), TandemHeart (TandemLife), and ProtekDuo (TandemLife). Each of these devices pumps blood from the vena cava and/or right atrium (RA) and ejects it into the PA, thereby bypassing the RV. Acute hemodynamic effects include reduction in right atrial pressure, increase in PA pressure, increase in left atrial pressure, and increase in cardiac output. Of note, when these devices are used in patients with biventricular failure, the increase in left atrial pressure may lead to pulmonary edema unless the additional LV preload can be offset by either diuresis/dialysis or direct LV unloading with an MCS device.
Impella RP is the only MCS device currently approved by the US Food and Drug Administration for the treatment of acute RV failure. The device utilizes a single 23-F femoral venous access for insertion of a preshaped pigtail catheter that houses an axial flow pump. The inlet portion of the catheter lies at the junction between the inferior vena cava and RA, and the outflow portion lies in the main PA. The safety and efficacy of Impella RP has been demonstrated in patients with RV shock attributable to RVI, post-LVAD implantation, and postcardiotomy. The RECOVER-RIGHT study included 30 such patients with acute RV failure treated with Impella RP; 18 patients had acute RV failure complicating implantable LVAD implantation. In this cohort, survival to 30 days or hospital discharge was 83.3%. In contrast, in the subset of patients treated with Impella RP for acute RV failure due to cardiotomy or acute MI, survival was 58.3%. The most common adverse events were bleeding and hemolysis.20
TandemHeart utilizes two 21-F cannulas inserted into either both femoral veins or one femoral vein and one internal jugular vein. The inflow cannula is placed in the RA, and the outflow cannula is placed in the PA. A centrifugal flow pump connects the two cannulas. The ProtekDuo incorporates the same centrifugal flow pump, but the inflow and outflow cannulas are combined into one double-lumen catheter that is inserted via the internal jugular vein. Both TandemHeart and ProtekDuo can be fitted with an oxygenator, which may be useful in the setting of RV failure with hypoxemia. The largest series reported outcomes of 46 patients treated with TandemHeart for a variety of indications.21 In-hospital mortality was 57% but was higher in patients with RV failure due to acute MI or post-LVAD implantation.
Indirect Right Ventricular Support
Venoarterial extracorporeal membrane oxygenation (VA-ECMO) utilizes a centrifugal pump to aspirate blood via a cannula inserted into the central venous circulation, pass it through a membrane oxygenator, and eject it into the central arterial circulation. Acute hemodynamic effects include reductions in both RV preload and RV cardiac output. Importantly, VA-ECMO also increases systemic arterial pressure and LV afterload. Particularly in the setting of biventricular failure, insertion of an additional MCS device or a left atrial/ventricular drain to unload the left ventricle may be necessary to prevent worsening LV failure and pulmonary edema. No clinical trials have been conducted to evaluate VA-ECMO for the treatment of acute RV failure, but the utility of this treatment strategy for a variety of clinical indications has been described in several case series.22-25
Biventricular Support
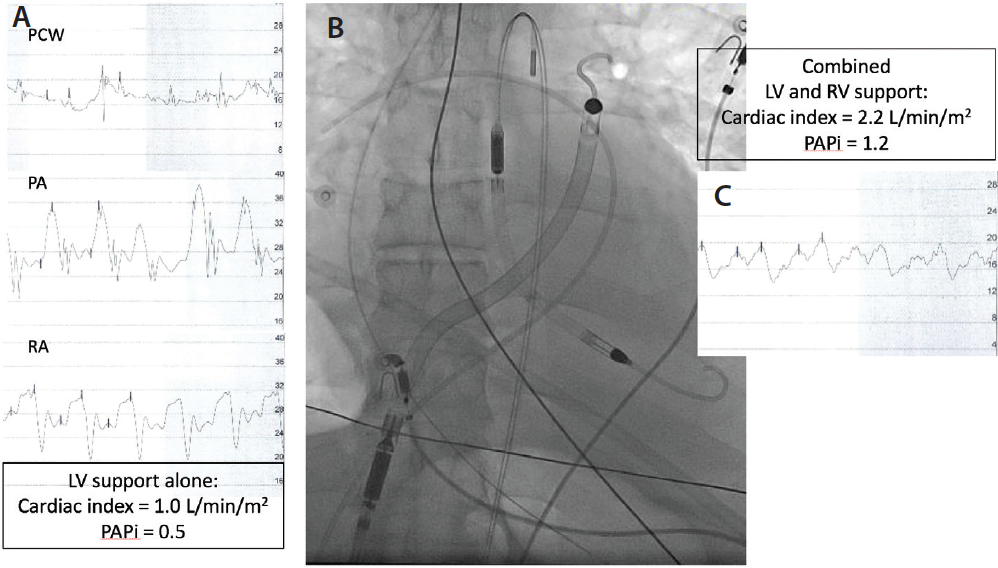
Figure 2. Invasive hemodynamic tracings of a patient who was initially treated with a percutaneous LVAD for LV failure (A). The patient became hypotensive, and based largely on the invasive hemodynamic data, an Impella RP device was inserted (B), resulting in improved cardiac output and reduced right atrial pressure (C). PCW, pulmonary capillary wedge.
Although VA-ECMO “bypasses” both the left and right ventricles, it has limited utility in the treatment of biventricular failure because the anticipated LV afterload will worsen LV hemodynamics without an additional MCS device to unload the LV. However, direct RV support devices in combination with either direct LV support devices (Impella, TandemHeart) or IABP have been used to successfully treat biventricular failure as a bridge to recovery, durable circulatory support, or cardiac surgery (Figure 2).26-31
CONCLUSION
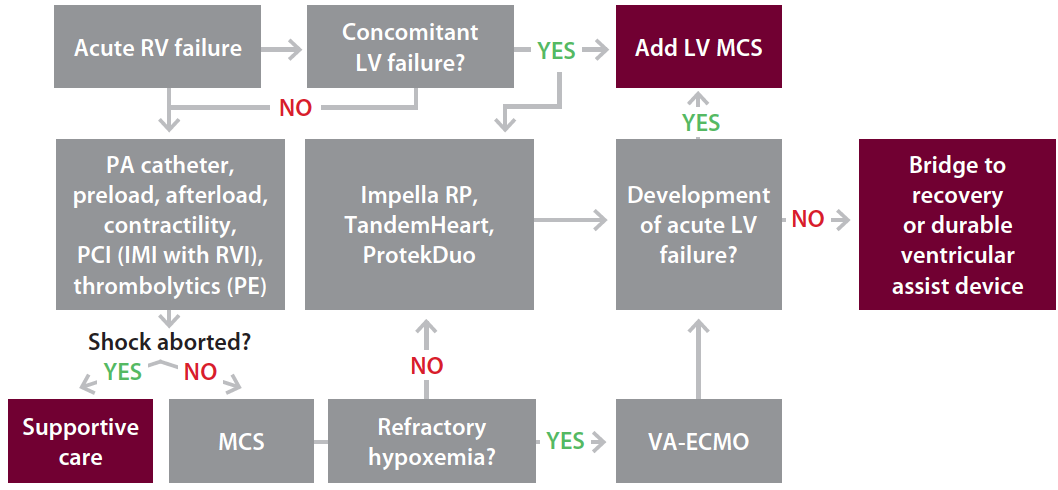
Figure 3. Proposed algorithm for management of acute RV failure. PCI, percutaneous coronary intervention; PE, pulmonary embolism.
Acute RV failure requires timely diagnosis, which is often guided by appropriate echocardiographic and invasive hemodynamic data. Immediate resuscitative efforts should target optimization of RV preload, afterload, and contractility and revascularization as clinically indicated. The need for MCS is determined by the degree of target organ perfusion and response to noninvasive therapies (Figure 3). The choice of MCS device depends on device availability and expertise of the treating institution and operator. If RV failure is associated with impaired oxygenation, a device equipped with a membrane oxygenator may be useful.
1. Rihal CS, Naidu SS, Givertz MM, et al. 2015 SCAI/ACC/HFSA/STS clinical expert consensus statement on the use of percutaneous mechanical circulatory support devices in cardiovascular care: Endorsed by the American Heart Association, the Cardiological Society of India, and Sociedad Latino Americana de Cardiologia Intervencion;
Affirmation of Value by the Canadian Association of Interventional Cardiology-Association Canadienne de Cardiologie d’intervention. J Am Coll Cardiol. 2015;65:e7-e26.
2. Goldstein JA, Barzilai B, Rosamond TL, et al. Determinants of hemodynamic compromise with severe right ventricular infarction. Circulation. 1990;82:359-368.
3. Goldstein JA, Tweddell JS, Barzilai B, et al. Importance of left ventricular function and systolic ventricular interaction to right ventricular performance during acute right heart ischemia. J Am Coll Cardiol. 1992;19:704-711.
4. Goldstein JA, Harada A, Yagi Y, et al. Hemodynamic importance of systolic ventricular interaction, augmented right atrial contractility and atrioventricular synchrony in acute right ventricular dysfunction. J Am Coll Cardiol. 1990;16:181-189.
5. Goldstein JA. Pathophysiology and management of right heart ischemia. J Am Coll Cardiol. 2002;40:841-853.
6. Basir MB, Schreiber T, Dixon S, et al. Feasibility of early mechanical circulatory support in acute myocardial infarction complicated by cardiogenic shock: the Detroit cardiogenic shock initiative [published online December 20, 2017]. Catheter Cardiovasc Interv.
7. Fincke R, Hochman JS, Lowe AM, et al. Cardiac power is the strongest hemodynamic correlate of mortality in cardiogenic shock: a report from the SHOCK trial registry. J Am Coll Cardiol. 2004;44:340-348.
8. Kormos RL, Teuteberg JJ, Pagani FD, et al. Right ventricular failure in patients with the HeartMate II continuous-flow left ventricular assist device: incidence, risk factors, and effect on outcomes. J Thorac Cardiovasc Surg. 2010;139:1316-1324.
9. Morine KJ, Kiernan MS, Pham DT, et al. Pulmonary artery pulsatility index is associated with right ventricular failure after left ventricular assist device surgery. J Card Fail. 2016;22:110-116.
10. Korabathina R, Heffernan KS, Paruchuri V, et al. The pulmonary artery pulsatility index identifies severe right ventricular dysfunction in acute inferior myocardial infarction. Catheter Cardiovasc Interv. 2012;80:593-600.
11. Hrymak C, Strumpher J, Jacobsohn E. Acute right ventricle failure in the intensive care unit: assessment and management. Can J Cardiol. 2017;33:61-71.
12. Kholdani CA, Fares WH. Management of right heart failure in the intensive care unit. Clin Chest Med. 2015;36:511-520.
13. Ventetuolo CE, Klinger JR. Management of acute right ventricular failure in the intensive care unit. Ann Am Thorac Soc. 2014;11:811-822.
14. Jacobs AK, Leopold JA, Bates E, et al. Cardiogenic shock caused by right ventricular infarction: a report from the SHOCK registry. J Am Coll Cardiol. 2003;41:1273-1279.
15. Bowers TR, O’Neill WW, Grines C, et al. Effect of reperfusion on biventricular function and survival after right ventricular infarction. N Engl J Med. 1998;338:933-940.
16. Haddad F, Doyle R, Murphy DJ, Hunt SA. Right ventricular function in cardiovascular disease, part II: pathophysiology, clinical importance, and management of right ventricular failure. Circulation. 2008;117:1717-1731.
17. Kearon C, Akl EA, Ornelas J, et al. Antithrombotic therapy for VTE disease: CHEST guideline and expert panel report. Chest. 2016;149:315-352.
18. McNamara MW, Dixon SR, Goldstein JA. Impact of intra-aortic balloon pumping on hypotension and outcomes in acute right ventricular infarction. Coron Artery Dis. 2014;25:602-607.
19. Goldstein JA, Kommuri N, Dixon SR. Left ventricular systolic dysfunction is associated with adverse outcomes in acute right ventricular infarction. Coron Artery Dis. 2016;27:277-286.
20. Anderson MB, Goldstein J, Milano C, et al. Benefits of a novel percutaneous ventricular assist device for right heart failure: the prospective RECOVER RIGHT study of the Impella RP device. J Heart Lung Transplant. 2015;34:1549-1560.
21. Kapur NK, Paruchuri V, Jagannathan A, et al. Mechanical circulatory support for right ventricular failure. JACC Heart Fail. 2013;1:127-134.
22. Belohlavek J, Rohn V, Jansa P, et al. Veno-arterial ECMO in severe acute right ventricular failure with pulmonary obstructive hemodynamic pattern. J Invasive Cardiol. 2010;22:365-369.
23. Corsi F, Lebreton G, Brechot N, et al. Life-threatening massive pulmonary embolism rescued by venoarterial-extracorporeal membrane oxygenation. Crit Care. 2017;21:76.
24. George B, Parazino M, Omar HR, et al. A retrospective comparison of survivors and non-survivors of massive pulmonary embolism receiving veno-arterial extracorporeal membrane oxygenation support. Resuscitation. 2018;122:1-5.
25. Suguta M, Hoshizaki H, Anno M, et al. Right ventricular infarction with cardiogenic shock treated with percutaneous cardiopulmonary support: a case report. Jpn Circ J. 1999;63:813-815.
26. Aghili N, Bader Y, Vest AR, et al. Biventricular circulatory support using 2 axial flow catheters for cardiogenic shock without the need for surgical vascular access. Circ Cardiovasc Interv. 2016;9.
27. Renard BM, Hanson ID, Goldstein JA. Severe mitral regurgitation and biventricular heart failure successfully treated with biventricular percutaneous axial flow pumps as a bridge to mitral valve surgery. Catheter Cardiovasc Interv. 2017;89:159-162.
28. Atwater BD, Nee LM, Gimelli G. Long-term survival using intra-aortic balloon pump and percutaneous right ventricular assist device for biventricular mechanical support of cardiogenic shock. J Invasive Cardiol. 2008;20:E205-207.
29. Rajagopal V, Steahr G, Wilmer CI, Raval NY. A novel percutaneous mechanical biventricular bridge to recovery in severe cardiac allograft rejection. J Heart Lung Transplant. 2010;29:93-95.
30. Nagy CD, Jumean MF, Pham DT, et al. Percutaneous circulatory support for biventricular failure. Circ Cardiovasc Interv. 2013;6:e12-14.
31. Kapur NK, Jumean M, Ghuloom A, et al. First successful use of 2 axial flow catheters for percutaneous biventricular circulatory support as a bridge to a durable left ventricular assist device. Circ Heart Fail. 2015;8:1006-1008.
Ivan D. Hanson, MD
Department of Cardiovascular Medicine
Beaumont Health System
Royal Oak, Michigan
ivan.hanson@beaumont.org
Disclosures: Speaker for Abiomed, Inc.
James A. Goldstein, MD
Department of Cardiovascular Medicine
Beaumont Health System
Royal Oak, Michigan
Disclosures: Consultant to Abiomed, Inc.
Advertisement
Advertisement
