Advertisement
Advertisement
January/February 2018
Technologies for Treating Left Atrial Decompression in Heart Failure
An overview of three unique devices that are currently in development, including data from early clinical trial experience and the potential advantages and pitfalls to be explored.
Dyspnea and exercise intolerance are fundamental clinical features of heart failure that are caused in part by resting or exercise-associated increases in left atrial pressure and consequent pulmonary vascular congestion. Therapies that aim to reduce total body volume and intravascular pressure have been associated with improved outcomes among patients who have heart failure with reduced ejection fraction (HFrEF) (ejection fraction [EF] < 40%), but not heart failure with preserved ejection fraction (HFpEF) (EF > 55%).1,2 This may be partly because the latter group is not necessarily total body volume overloaded, but exhibits exertional increases in left atrial pressure.3,4 Accordingly, more selective reductions in left atrial pressure, if achievable, may confer symptomatic benefits without the potentially negative sequelae of pharmacologic volume removal and additionally mitigate concerns regarding drug adherence and resistance.
Balloon septostomy has been reported in cases of recalcitrant heart failure and hypoxemia,5 and the utilization of a fenestrated Amplazter Septal Occluder (Abbott Vascular, formerly St. Jude Medical) to maintain a durable Fontan fenestration and interatrial communication was described more than 15 years ago.6 Mechanical devices that aim to reduce left atrial pressure have been developed and evaluated in HFpEF and HFrEF patients. This article provides an update on three such devices, along with early clinical experience and outlook for the long term.
IASD SYSTEM
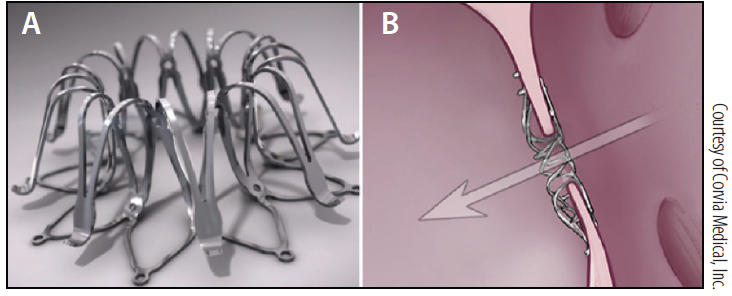
Figure 1. The IASD system. Once deployed, there is a connection between the right and left atrium, enabling flow dictated by the pressure gradient, left to right atrium (A). The device is composed of two nitinol discs around a central fenestration (B).
The largest clinical experience to date exists with the IASD (interatrial shunt device) system developed by Corvia Medical, Inc. This nitinol device is composed of a left and right atrial disc (19-mm outer diameter), with an 8-mm communication. The device is deployed percutaneously via a sheath (16 F) in the femoral vein, with fluoroscopic and intracardiac or transesophageal echocardiographic guidance. The device is deployed after transseptal puncture of the mid–fossa ovalis, positioning the delivery catheter into the left atrium and deploying the left atrial disc, retracting and apposing this disc to the atrial septum, verifying the right atrial location of the delivery catheter, then deploying the right atrial disc such that the device is secured across the atrial septum (Figure 1).
Initial human experience was published in 2014.7 Eleven patients were treated (EF > 45%, all with New York Heart Association [NYHA] III/IV heart failure and pulmonary capillary wedge pressure [PCWP] > 15 mm Hg at rest or > 25 mm Hg with exercise) in a pilot study. The device was successfully implanted in all patients. At 30 days, PCWP had decreased in 10 of 11 patients (mean, 19 ± 5 mm Hg at baseline and 14 ± 3 mm Hg at 30 days; P < .01), most (7/11) had improved by at least one NYHA class, and there was significant increase in 6-minute walk distance. These observations were extended in the REDUCE LAP-HF trial, a phase 1, open-label, single-arm trial in which 66 patients underwent the procedure.8 Implantation success was high (64/66; 97%), without major periprocedural adverse events. At 6-month follow-up, most patients had reductions in PCWP at rest and with exertion (52% and 58%, respectively), along with significant improvements in NYHA class and 6-minute walk distance (from 313 to 345 m; P = .023). These changes occurred in tandem with sustained left-to-right interatrial flow and pulmonary to systemic blood flow ratio (Qp:Qs ~1.3:1). At 1-year follow-up, there was a persistent and significant reduction in the pressure difference between the left and right atrium at rest and with exertion, again with improved NYHA class and 6-minute walk distance, as well as sustained shunt Qp/Qs ratio. In addition, fewer heart failure hospitalizations were observed in the year after implantation (mean, 1.5–0.8 per year; P < .05).9,10
Based on this study, the IASD system received CE Mark approval for the treatment of HFpEF and heart failure with midrange ejection fraction (HFmrEF) (EF, 40%–55%).2 Patients have been randomized in the United States, Australia, and Europe (N = 44) in the REDUCE LAP-HF I trial. One-month outcomes were recently reported; there were no periprocedural adverse events, and treated patients exhibited greater reductions in exercise-associated PCWP.11 A larger-scale trial (REDUCE LAP-HF II) of the IASD in HFpEF/HFmrEF patients has commenced recruitment, with an enrollment goal of 380 patients (NCT03088033).
V-WAVE DEVICE
The V-Wave device (V-Wave Ltd.) includes an hourglass-shaped nitinol frame that is partially covered with expanded polytetrafluoroethylene (ePTFE) that serves as an anchor for a porcine trileaflet pericardial valve. The valve is designed to prevent right-to-left atrial flow (Figure 2). It is implanted via the femoral venous approach (14 F) under fluoroscopic and echocardiographic guidance. After transseptal puncture in the center of the fossa ovalis, the waist of the hourglass (5.1-mm inner diameter) is placed across the fossa, securing the device in place.
Preclinical experience with the V-Wave device was achieved in a large animal (ovine) model, with induction of heart failure.12 Animals (N = 21) were randomized to placement of the V-Wave device or a sham procedure. Twelve weeks after randomization, animals that received the V-Wave device had lower mean left atrial and right atrial pressures (25 vs 14 mm Hg; P < .001) and higher mean EF (17% vs 46%; P < .001), but comparable mean left ventricular diastolic dimensions (5.2 vs 5.4 cm; P = .91). Resting Qp:Qs was approximately 1.2:1.
First-in-human experience was reported in 10 patients with HFrEF (all NYHA III; mean EF, 25%). The device was successfully implanted in all enrolled patients (N = 10), and nine survived to 3-month follow-up. At 3 months, there were significant reductions in mean PCWP (from 23 to 17 mm Hg; P = .035) and 6-minute walk distance (from 244 to 310 m; P = .016), with most (8/9) experiencing improved NYHA class.13,14 At 1-year follow-up, resting shunt fraction had declined from a mean of 1.2:1 to 1.1:1, with 14% of patients having no interatrial flow. This occurred in conjunction with pannus thickening of the bioprosthetic leaflets and lumen loss, which prompted the creation of a second-generation device without the one-way valve component, extended the ePTFE coating, and included a hood to prevent potential thromboemboli from apposing the right atrial side of the implant. This version of the device has been evaluated histologically, and late lumen loss has not been observed at 6 months.15
ATRIAL FLOW REGULATOR
Occlutech International AB has developed an atrial flow regulator (AFR) that is a nitinol mesh device composed of two flat discs and a 1- to 2-mm connecting neck with a central fenestration that enables bidirectional flow (Figure 3). It is manufactured in fenestration sizes of 6, 8, or 10 mm and is delivered via femoral venous approach with a 10- to 12-F sheath after an atrial septostomy.
The first clinical utilization of the device followed a compassionate use approval from the US Food and Drug Administration. The patient was a 54-year-old woman with severe and irreversible pulmonary artery hypertension. Implantation was successful and was associated with right-to-left shunting and a corresponding decrease in arterial saturation (from 95% to 89%). She reported functional improvement at 6 weeks.16 This experience was later extended to 12 patients. There were no periprocedural adverse events. Each patient reported symptomatic improvement, and 6-minute walk distance improved (from 377 to 423 m; P < .01) at a mean follow-up of approximately 6 months.17
A human international trial is forthcoming via the PRELIEVE trial. It is an open-label, nonrandomized trial including up to 30 patients with symptomatic heart failure (HFpEF or HFrEF) and a heart failure admission in the past 12 months (NCT03030274).
LONG-TERM CONSIDERATIONS FOR INTERATRIAL SHUNTING
The theoretical consequences of an iatrogenic left-to-right atrial shunt tend to stimulate rich conversation when considering these technologies. Among the theoretical foreseeable outcomes are elevation in right-sided pressures, with related chamber dilation and symptoms of right heart failure. Although congenital literature suggests small shunts (< 1.3:1) are generally tolerable for decades,18 the adult heart failure population is clearly a different phenotype. Reported experience with the IASD system in heart failure patients has revealed nonsignificant increases in right atrial and ventricular volumes at 6 months after implantation, which did not progress at 12-month follow-up and have not occurred in tandem with a decrement in right ventricular function.8,10 There were no increases in right-sided volumes in preclinical and early clinical trials of the V-Wave device—perhaps explained by the different flow characteristics, shunt diameter, and/or the different patient populations studied.8,12,13 As shunt fraction is theoretically a modifiable feature related to shunt diameter and intracardiac pressure,19 it is conceivable that it could be tailored to the underlying cardiac disorder at the time of implantation or amended (even closed) should right heart failure develop.
Additionally, because atrial arrhythmias are a common manifestation of unrepaired congenital atrial septal defects20,21 and have been observed with greater frequency in the months after implantation of other atrial septal devices,22,23 ongoing surveillance is warranted as these trials unfold. This will be important, and potentially complex, given that atrial arrhythmias are also common in heart failure, which itself may contribute to the development of arrhythmias. Paradoxical embolism represents a third potential concern. Fortunately, these events are rare among individuals with congenital interatrial shunts and have not been observed to date with the aforementioned devices.24
CONCLUSION
This class of technologies has been associated with improved functional status and hemodynamics in early trials (Table 1). These findings allude to an exciting future for the device-based treatment of heart failure across pathophysiologic spectrums, independent of the constraints of pharmacologic therapy.
1. Yancy CW, Jessup M, Bozkurt B, et al. 2013 ACCF/AHA guideline for the management of heart failure: a report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines. J Am Coll Cardiol. 2013;62:e147-239.
2. Nadruz W Jr, West E, Santos M, et al. Heart failure and midrange ejection fraction: implications of recovered ejection fraction for exercise tolerance and outcomes. Circ Heart Fail. 2016;9:e002826.
3. Maeder MT, Thompson BR, Htun N, Kaye DM. Hemodynamic determinants of the abnormal cardiopulmonary exercise response in heart failure with preserved left ventricular ejection fraction. J Card Fail. 2012;18:702-710.
4. Borlaug BA, Nishimura RA, Sorajja P, et al. Exercise hemodynamics enhance diagnosis of early heart failure with preserved ejection fraction. Circ Heart Fail. 2010;3:588-595.
5. Seib PM, Faulkner SC, Erikson CC, et al. Blade and balloon atrial septostomy for left heart decompression in patients with severe ventricular dysfunction on extracorporeal membrane oxygenation. Catheter Cardiovasc Interv. 1999;46:179-186.
6. Amin Z, Danford DA, Pedra CA. A new Amplatzer device to main patency of fontan fenestrations and atrial septal defects. Catheter Cardiovasc Interv. 2002;57:246-251.
7. Sondergaard L, Reddy V, Kaye D, et al. Transcatheter treatment of heart failure with preserved or mildly reduced ejection fraction using a novel interatrial implant to lower left atrial pressure. Eur J Heart Fail. 2014;16:796-801.
8. Hasenfuss G, Hayward C, Burkhoff D, et al. A transcatheter intracardiac shunt device for heart failure with preserved ejection fraction (REDUCE LAP-HF): a multicentre, open-label, single-arm, phase 1 trial. Lancet. 2016;387:1298-1304.
9. Malek F, Neuzil P, Gustafsson F, et al. Clinical outcome of transcatheter treatment of heart failure with preserved or mildly reduced ejection fraction using a novel implant. Int J Cardiol. 2015;187:227-228.
10. Kaye DM, Hasenfuss G, Neuzil P, et al. One-year outcomes after transcatheter insertion of an interatrial shunt device for the management of heart failure with preserved ejection fraction. Circ Heart Fail. 2016;9.
11. Feldman T, Mauri L, Kahwash R, et al. A transcatheter interatrial shunt device for the treatment of heart failure with preserved ejection fraction (REDUCE LAP-HF I): a phase 2, randomized, sham-controlled trial [published online November 15, 2017]. Circulation.
12. Eigler NL, del Rio CL, Verheye S, et al. Cardiac unloading with an implantable interatrial shunt in heart failure: serial observations in an ovine model of ischemic cardiomyopathy. Structural Heart. 2017;1:40-48.
13. Del Trigo M, Bergeron S, Bernier M, et al. Unidirectional left-to-right interatrial shunting for treatment of patients with heart failure with reduced ejection fraction: a safety and proof-of-principle cohort study. Lancet. 2016;387:1290-1297.
14. Amat-Santos IJ, Bergeron S, Bernier M, et al. Left atrial decompression through unidirectional left-to-right interatrial shunt for the treatment of left heart failure: first-in-man experience with the V-Wave device. EuroIntervention. 2015;10:1127-1131.
15. Stone GW R-CJ, Amat-Santos IJ, Ben Gal T, et al. Interatrial shunting for heart failure: the V-Wave shunt. Presented at Transcatheter Therapeutics (TCT); October 31, 2017; Denver, Colorado.
16. Patel MB, Samuel BP, Girgis RE, et al. Implantable atrial flow regulator for severe, irreversible pulmonary arterial hypertension. EuroIntervention. 2015;11:706-709.
17. Rajeshkumar R, Pavithran S, Sivakumar K, Vettukattil JJ. Atrial septostomy with a predefined diameter using a novel occlutech atrial flow regulator improves symptoms and cardiac index in patients with severe pulmonary arterial hypertension [published online September 1, 2017]. Catheter Cardiovasc Interv.
18. Warnes CA, Williams RG, Bashore TM, et al. ACC/AHA 2008 guidelines for the management of adults with congenital heart disease: executive summary: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines (writing committee to develop guidelines for the management of adults with congenital heart disease). Circulation. 2008;118:2395-2451.
19. Kaye D, Shah SJ, Borlaug BA, et al. Effects of an interatrial shunt on rest and exercise hemodynamics: results of a computer simulation in heart failure. J Card Fail. 2014;20:212-221.
20. Berger F, Vogel M, Kramer A, et al. Incidence of atrial flutter/fibrillation in adults with atrial septal defect before and after surgery. Ann Thorac Surg. 1999;68:75-78.
21. Konstantinides S, Geibel A, Olschewski M, et al. A comparison of surgical and medical therapy for atrial septal defect in adults. N Engl J Med. 1995;333:469-473.
22. Sondergaard L, Kasner SE, Rhodes JF, et al. Patent foramen ovale closure or antiplatelet therapy for cryptogenic stroke. N Engl J Med. 2017;377:1033-1042.
23. Komar M, Przewlocki T, Olszowska M, et al. Conduction abnormality and arrhythmia after transcatheter closure of atrial septal defect. Circ J. 2014;78:2415-2421.
24. Saver JL, Carroll JD, Thaler DE, et al. Long-term outcomes of patent foramen ovale closure or medical therapy after stroke. N Engl J Med. 2017;377:1022-1032.
Scott M. Lilly, MD, PhD
Assistant Professor, Interventional Cardiology
Medical (Interventional) Director, Structural Heart Disease
Program
Division of Cardiovascular Medicine
The Ohio State University
Columbus, Ohio
scott.lilly@osumc.edu
Disclosures: None.
Daniel Burkhoff, MD, PhD
Director
Heart Failure, Hemodynamics, and Mechanical Circulatory Support
Research
Cardiovascular Research Foundation
New York, New York
Disclosures: Consultant to Corvia Medical, Inc.
Advertisement
Advertisement



