Advertisement
Advertisement
January/February 2018
Selection of Hemodynamic Support: An Approach for Coronary Interventions in Shock and High-Risk PCI
The role of hemodynamic support devices in emergent cardiogenic shock and elective high-risk percutaneous coronary intervention.
The majority of patients with cardiomyopathy and heart failure have an ischemic substrate. Although some patients present with acute myocardial infarction (AMI) with or without cardiogenic shock as an etiology of acute and then chronic progressive heart failure, others have more stable coronary disease, resulting in progressive dilation and cardiomyopathy, which leads to chronic heart failure. In both scenarios, the natural history of heart failure can be improved with optimal use of hemodynamic support devices. Studies to date, which are primarily observational but also randomized, have provided increasing support for the use of hemodynamic support devices for both AMI with cardiogenic shock and high-risk percutaneous coronary intervention (PCI) in patients with large ischemic burden.
In both scenarios, supported interventions can either acutely or chronically improve the substrate for heart failure, impacting the natural history and quality of life for these patients. The goal of hemodynamic support in these situations is to decrease myocardial workload, unload the left ventricle (LV), improve cardiac output, and maintain adequate coronary perfusion during revascularization procedures. The most commonly available percutaneous left ventricular assist devices used for hemodynamic support are the intra-aortic balloon pump (IABP), Impella 2.5/CP/5.0 (Abiomed, Inc.), TandemHeart (TandemLife), and extracorporeal membrane oxygenation (ECMO). As TandemHeart is infrequently utilized percutaneously, this article will primarily focus on the role of IABP, Impella, and ECMO for hemodynamic support in emergent cardiogenic shock PCI and elective “high-risk” PCI.
CARDIOGENIC SHOCK
Cardiogenic shock is a state of reduced cardiac output due to primary cardiac dysfunction leading to inadequate tissue perfusion and hypotension.1 Frequently used diagnostic criteria include systolic blood pressure < 90 mm Hg (systolic blood pressure > 90 mm Hg with support) and end-organ hypoperfusion (urine output < 30 mL/h or cool extremities). Hemodynamic criteria include cardiac index of ≤ 2.2 L/min/m2 and elevated filling pressures (pulmonary capillary wedge pressure ≥ 15 mm Hg).2 Cardiogenic shock after ST-segment elevation myocardial infarction (STEMI) continues to be associated with poorer outcomes and higher in-hospital mortality.
In our analysis of national administrative databases, the overall incidence of cardiogenic shock in STEMI patients was 8%, with an in-hospital mortality of 39%.3 In-hospital mortality was > 50% in patients aged ≥ 75 years.3 The maladaptive vicious cycle of vasoconstriction, neurohormonal activation, and salt/water retention that develops during cardiogenic shock further compromises hemodynamics. Percutaneous ventricular assist devices have been utilized and studied most in this subset of patients. An ideal assist device in this scenario should have fast implantation times, provide adequate hemodynamic support, unload the LV, and have minimal vascular complications.
Intra-Aortic Balloon Pump
The most commonly utilized technique for percutaneous hemodynamic support during cardiogenic shock is the IABP, which is used in > 50% of STEMI patients with cardiogenic shock in United States hospitals.3 It can be inserted relatively quickly, requires an 8-F arterial access (7.5 F for sheathless), and is placed in the descending aorta just below the left subclavian artery. The IABP is inflated during diastole and displaces blood in the aorta. It works by augmenting diastolic aortic pressure, increasing systemic mean pressure, reducing LV and aortic systolic pressures, and improving coronary perfusion. The drop in afterload improves stroke volume, with estimates of increasing cardiac output by roughly 0.5 to 1 L/min (Figure 1).
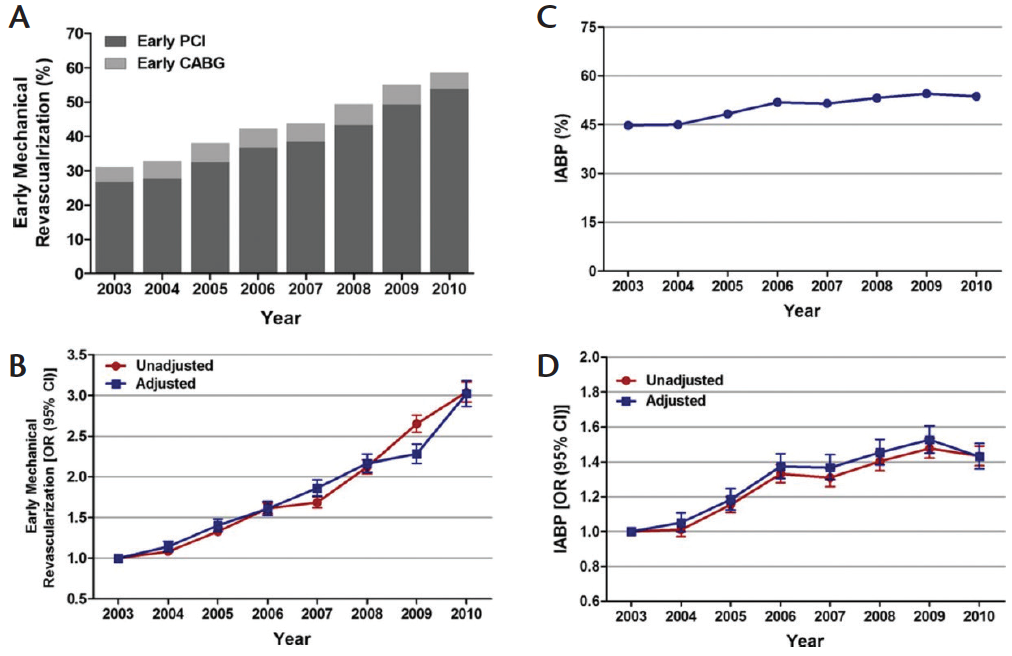
Figure 2. Trends in early mechanical revascularization and IABP use in patients with STEMI complicated by cardiogenic shock. Early mechanical revascularization was defined as PCI or coronary artery bypass grafting (CABG) within 24 hours of admission; P for trend < .001 (A). Trends in early mechanical revascularization presented as unadjusted and adjusted ORs and 95% CIs for each year relative to 2003 (reference year). Regression model adjusted for demographics, hospital characteristics, 29 Elixhauser Comorbidity Index and other clinically relevant comorbidities, and presentation (B). IABP (%) was calculated as the number of patients undergoing IABP placement divided by the number of patients with STEMI complicated by cardiogenic shock per year X 100; P for trend < .001 (C). Trends in IABP use presented as unadjusted and adjusted ORs and 95% CIs for each year relative to 2003 (reference year). Regression model adjusted for demographics, hospital characteristics, 29 Elixhauser Comorbidity Index and other clinically relevant comorbidities, and presentation (D). Reprinted from Kolte D, Khera S, Aronow WS, et al. Trends in incidence, management, and outcomes of cardiogenic shock complicating ST-elevation myocardial infarction in the United States. J Am Heart Assoc. 2014;3:e000590.
Despite its widespread utilization in conjunction with percutaneous revascularization (Figure 2) for STEMI patients in cardiogenic shock, the data for routine use have been disappointing. Older data from the National Registry of Myocardial Infarction 2 reported lower in-hospital mortality when the IABP was instituted with thrombolytics but not when used with PCI.4 The more recent IABP-SHOCK trials, especially the IABP-SHOCK II trial that randomized 600 patients with cardiogenic shock complicating AMI to IABP support (n = 301) and no IABP support (n = 299), reported no differences between the two groups in terms of 30-day mortality (relative risk with IABP, 0.96; 95% confidence interval [CI], 0.79–1.17; P = .69).5,6 The mortality reported in the trial was around 40%, leading to speculation that a majority of patients had hemodynamically mild to moderately severe cardiogenic shock.
In totality, the data are weak to support “routine” utilization of IABP in all-comer cardiogenic shock patients undergoing PCI. Accordingly, the 2015 European Society of Cardiology guidelines have moved routine IABP use to a class III recommendation based on lack of apparent benefit and potential stroke risk.7 However, it should be emphasized that in patients with mechanical complications (ventricular septal rupture, acute mitral regurgitation), IABP should be employed and urgent surgical consultation should be sought. Ease of implantation, fewer vascular complications, familiarity with device operation by lab personnel, and cost make it the most accessible percutaneous left ventricular assist device in the market. Complications of IABP use include acute limb ischemia, hemolysis, balloon rupture, stroke, and death. IABP use is contraindicated in patients with severe peripheral artery disease and greater than moderate aortic regurgitation.
Impella
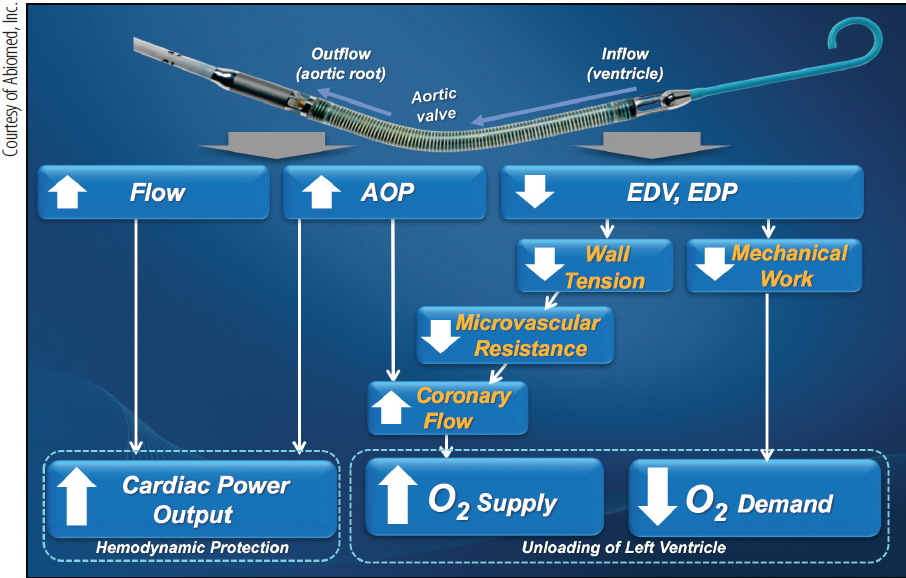
Figure 3. Hemodynamic effects of Impella support. AOP, aortic pressure; EDP, left ventricular end-diastolic pressure; EDV, left ventricular end-diastolic volume.
Impella is a minimally invasive percutaneous left ventricular assist device that is inserted via a 12-F (Impella 2.5, 2.5-L flow), 14-F (Impella CP, 3.3-L flow), and 21-F (Impella 5.0, 5-L flow) sheath. Impella 2.5 and CP are implanted in the catheterization laboratories via femoral or axillary routes. Impella is an axial flow pump placed across the aortic valve that propels blood from the LV (via the inflow cannula) to the aorta (via the outflow cannula). This increases the forward flow directly, which provides direct unloading of the LV (by decreasing the left ventricular end-diastolic pressure and volume) and increases the mean arterial and coronary perfusion pressures (Figure 3).
The ISAR-SHOCK trial was a small study comparing IABP (n = 13) to Impella 2.5 (n = 12) that demonstrated greater improvement in cardiac index 30 minutes after implantation of Impella versus IABP.8 The mortality rates were 46% in both groups at 30 days. The recent IMPRESS trial, which randomized patients with cardiogenic shock to Impella CP (n = 24) or IABP (n = 24), demonstrated no differences in mortality at 30 days (46% for Impella CP vs 50% for IABP; P = .92) and 6 months (50% for both groups; P = .923) between the two groups.9 Clearly, Impella provides a better hemodynamic profile and improved cardiac output when compared with IABP; however, there appears to be a lack of mortality benefit, which is presumed to be due to the small sample size and timing of initiation of mechanical support.
In this regard, the USPELLA registry reported a 63% greater in-hospital survival rate if Impella support was initiated prior to revascularization of the infarct-related artery in patients with cardiogenic shock (odds ratio [OR], 0.37; 95% CI, 0.17–0.79; P = .01).10 Initiation of mechanical circulatory support prior to revascularization leads to unloading of the LV; concomitant decrease in wall stress and associated oxygen demand may improve outcomes by improving ischemia during revascularization (and reperfusion), maintaining higher TIMI (thrombolysis in myocardial infarction) flow postprocedure, and ensuring stent patency. This forms the basis of the ongoing Door to Unloading With Impella CP System in Acute Myocardial Infarction trial.
Impella is associated with greater vascular complications (larger sheath size) and requires technical expertise and time compared with IABP insertion. However, most experienced centers can implant the Impella device in < 10 minutes compared to < 5 minutes for IABP. Complications of Impella include hemolysis and acute limb ischemia. Impella is contraindicated for use in patients with severe aortic stenosis, presence of LV thrombus, mechanical aortic valve, severe peripheral artery disease, more than moderate aortic valve insufficiency, and ventricular septal defect, although it has been used in the latter situation on occasion.
Extracorporeal Membrane Oxygenation
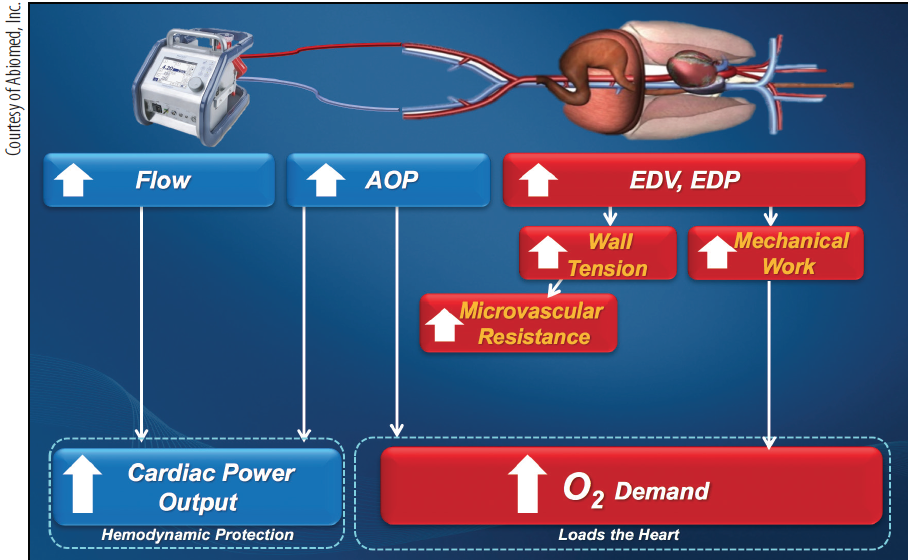
Figure 4. Hemodynamic effects of ECMO. AOP, aortic pressure; EDP, left ventricular end-diastolic pressure; EDV, left ventricular end-diastolic volume.
Venoarterial ECMO is a percutaneously inserted cardiopulmonary bypass with an 18- to 21-F inflow cannula placed in the right atrium (via femoral vein) and outflow cannula placed in the aorta (via large-bore 15- to 22-F catheter[s] in the femoral or axillary artery). The blood from the inflow cannula passes through a heat exchanger and oxygenator system and is then released into the arterial outflow cannula. Benefits include improved cardiac output (> 4.5 L/min), assisting both the right ventricle and LV, and bypass of the pulmonary circulation for oxygenation. These hemodynamic benefits come at the price of reduced ventricular loading, increased LV wall stress, and myocardial oxygen demand. Similar to bypass circuits in the operating room, the LV will be distended unless concomitant unloading occurs. Percutaneously, this can be achieved with Impella or IABP placement (Figure 4).
Small, observational, and often single-center studies have reported improved outcomes with ECMO in patients with profound cardiogenic shock due to acute coronary syndromes.11-14 A prospective observational study in STEMI-cardiogenic shock patients from Taiwan reported a 30-day survival of 60.9% on ECMO plus IABP versus 28% in patients on IABP support alone.11 In our opinion, ECMO should be reserved for patients with hypoxemic cardiogenic shock or biventricular failure, particularly when native heart recovery is unlikely. IABP or Impella support (to unload the LV) in conjunction with ECMO can be utilized in patients with profound hypoxemic cardiogenic shock at experienced centers.15,16 ECMO is associated with higher vascular complications, limb ischemia, and acute kidney injury and often requires antegrade percutaneous access to provide distal perfusion to the limb.
HIGH-RISK PCI
There has been a paradigm shift in the characteristics of lesions and patients encountered in modern-day catheterization laboratories. Improvement in stent design and pharmacotherapy has led to a decline in restenosis rates. This, coupled with an aging patient population with extensive comorbidities referred for PCI after surgical turndown, has led to an increased number of high-risk PCIs. “High risk” pertains to patient characteristics (eg, advanced age, comorbidities, renal insufficiency, and/or prohibitive surgical revascularization), anatomic/lesion characteristics (eg, multivessel disease, left main disease, and/or complex/calcified/chronic total occlusions), and baseline cardiac function (eg, elevated LV filling pressures and/or depressed LV function), either in combination or independent of each other after extensive individual patient evaluation. The most commonly encountered high-risk PCI situation is a patient with low ejection fraction (< 30%) and/or revascularization of a vessel supplying > 50% of the myocardium, in whom it is hoped revascularization will improve ejection fraction, quality of life, and incidence of repeat hospitalizations for heart failure.
In patients with impaired LV contractile reserve, profound negative hemodynamic changes may ensue with repeated contrast injections, transient occlusion with balloon inflations, manipulation of equipment in the coronary tree, or use of atherectomy devices. Mechanical circulatory support using IABP, Impella, or ECMO can be of paramount importance in this subset of patients not only to facilitate safe PCI but also to optimize the chances of improved cardiac function.
The BCIS-1 study was the first randomized trial that demonstrated a 34% relative risk reduction (OR, 0.66; 95% CI, 0.44–0.98; P = .039) in all-cause mortality at a median of 51 months in patients undergoing high-risk PCI with elective IABP implantation. Although not the primary endpoint, the trial also showed a reduction in procedural ischemic complications in patients randomized to routine IABP.17 The PROTECT II trial randomized patients with complex multivessel coronary artery disease or unprotected left main disease with depressed left ventricular ejection fraction (LVEF) to either IABP (n= 226) or Impella 2.5 (n = 226) support during elective high-risk PCI. The primary outcome (major adverse events at 30 days) was no different between the two groups (Impella 2.5, 35.1% vs IABP, 40.1%; P = .227 in the intention-to-treat population and Impella 2.5, 34.3% vs IABP, 42.2%; P = .092 in the per-protocol population). However, by 90 days, lower major adverse events were seen in the Impella 2.5 group in the per-protocol population (Impella 2.5, 40% vs IABP, 51%; P = .023).18 Furthermore, patients with three-vessel disease and LVEF ≤ 30% had significantly lower major adverse events at 90 days after Impella 2.5 support in a PROTECT II substudy.19 Patients enrolled in the PROTECT II trial were sicker and had more comorbidities, including diabetes, stroke, previous PCI, and coronary artery bypass graft surgery, compared with the BCIS-1 population.
Perhaps the biggest benefit shown in the PROTECT II trial was that both LVEF and functional status/quality of life at 90 days improved with Impella and IABP, indicating that high-risk multivessel revascularization has important benefits in terms of remodeling and improving the heart failure state. Clearly, there is a role for using IABP or Impella support in elective cases with low ejection fraction undergoing high-risk PCI to impact the natural history in patients most at risk for progressive reduction in heart failure if left untreated. Although ECMO has been utilized in patients undergoing high-risk PCI,20 technical challenges, issues of ventricular loading, and vascular complications preclude its widespread acceptance for elective procedures. In addition, the increased afterload that comes with ECMO would be theoretically disadvantageous during PCI, promoting further ischemia during the intervention.
CONCLUSION
Heart failure is a global epidemic, only growing in incidence and prevalence despite our best efforts. As a result, we should be acutely focused on mechanisms to reduce this incidence. Ischemic cardiomyopathy, which is still the majority of new heart failure diagnoses, is caused by AMI that results in acute shock or progressive remodeling over time despite our best medications or by chronic stable coronary disease and global ischemia with progressive remodeling despite medications. Both of these scenarios can be impacted mechanically by the timely institution of PCI, thereby breaking the cycle of progressive remodeling and perhaps obviating the need for polypharmacy.
In patients presenting with AMI and cardiogenic shock, supported PCI makes sense and is backed by some data, specifically if support is initiated quickly before PCI. In patients with multivessel disease who are poor candidates for coronary artery bypass grafting, supported PCI makes sense, with the aim to revascularize all ischemic territories. As these hemodynamic support devices become more commonplace, experience grows, and data accrues, we hopefully will see a decline in the incidence of heart failure and improved quality of life for these patients, with resultant improved prognosis.
1. van Diepen S, Katz JN, Albert NM, et al. Contemporary management of cardiogenic shock—a scientific statement from the American Heart Association. Circulation. 2017;136:e232-e268.
2. Hochman JS, Sleeper LA, Webb JG, et al. Early revascularization in acute myocardial infarction complicated by cardiogenic shock: SHOCK investigators: should we emergently revascularize occluded coronaries for cardiogenic shock. N Engl J Med. 1999;341:625-634.
3. Kolte D, Khera S, Aronow WS, et al. Trends in incidence, management, and outcomes of cardiogenic shock complicating ST-elevation myocardial infarction in the United States. J Am Heart Assoc. 2014;3:e000590.
4. Barron HV, Every NR, Parsons LS, et al. The use of intra-aortic balloon counterpulsation in patients with cardiogenic shock complicating acute myocardial infarction: data from the National Registry of Myocardial Infarction 2. Am Heart J. 2001;141:933-939.
5. Prondzinsky R, Lemm H, Swyter M, et al. Intra-aortic balloon counterpulsation in patients with acute myocardial infarction complicated by cardiogenic shock: the prospective, randomized IABP SHOCK trial for attenuation of multiorgan dysfunction syndrome. Crit Care Med. 2010;38:152-160.
6. Thiele H, Zeymer U, Neumann FJ, et al. Intraaortic balloon support for myocardial infarction with cardiogenic shock. N Engl J Med. 2012;367:1287-1296.
7. Roffi M, Patrono C, Collet JP, et al. 2015 ESC guidelines for the management of acute coronary syndromes in patients presenting without persistent ST-segment elevation: task force for the management of acute coronary syndromes in patients presenting without persistent st-segment elevation of the European Society of Cardiology (ESC). Eur Heart J. 2016;37:267-315.
8. Seyfarth M, Sibbing D, Bauer I, et al. A randomized clinical trial to evaluate the safety and efficacy of a percutaneous left ventricular assist device versus intra-aortic balloon pumping for treatment of cardiogenic shock caused by myocardial infarction. J Am Coll Cardiol. 2008;52:1584-1588.
9. Ouweneel DM, Eriksen E, Sjauw KD, et al. Percutaneous mechanical circulatory support versus intra-aortic balloon pump in cardiogenic shock after acute myocardial infarction. J Am Coll Cardiol. 2017;69:278-287.
10. O’Neill WW, Schreiber T, Wohns DH, et al. The current use of Impella 2.5 in acute myocardial infarction complicated by cardiogenic shock: results from the USpella Registry. J Interv Cardiol. 2014;27:1-11.
11. Sheu JJ, Tsai TH, Lee FY, et al. Early extracorporeal membrane oxygenator-assisted primary percutaneous coronary intervention improved 30-day clinical outcomes in patients with ST-segment elevation myocardial infarction complicated with profound cardiogenic shock. Crit Care Med. 2010;38:1810-1817.
12. Sattler S, Khaladj N, Zaruba MM, et al. Extracorporal life support (ECLS) in acute ischaemic cardiogenic shock. Int J Clin Pract. 2014;68:529-531.
13. Sakamoto S, Taniguchi N, Nakajima S, Takahashi A. Extracorporeal life support for cardiogenic shock or cardiac arrest due to acute coronary syndrome. Ann Thorac Surg. 2012;94:1-7.
14. Chung SY, Tong MS, Sheu JJ, et al. Short-term and long-term prognostic outcomes of patients with ST-segment elevation myocardial infarction complicated by profound cardiogenic shock undergoing early extracorporeal membrane oxygenator-assisted primary percutaneous coronary intervention. Int J Cardiol. 2016;223:412-417.
15. Pappalardo F, Schulte C, Pieri M, et al. Concomitant implantation of Impella on top of veno-arterial extracorporeal membrane oxygenation may improve survival of patients with cardiogenic shock. Eur J Heart Fail. 2017;19:404-412.
16. Karatolios K, Chatzis G, Markus B, et al. Biventricular unloading in patients with refractory cardiogenic shock. Int J Cardiol. 2016;222:247-252.
17. Perera D, Stables R, Clayton T, et al. Long-term mortality data from the balloon pump-assisted coronary intervention study (BCIS-1): a randomized, controlled trial of elective balloon counterpulsation during high-risk percutaneous coronary intervention. Circulation. 2013;127:207-212.
18. O’Neill WW, Kleiman NS, Moses J, et al. A prospective, randomized clinical trial of hemodynamic support with Impella 2.5 versus intra-aortic balloon pump in patients undergoing high-risk percutaneous coronary intervention: the PROTECT II study. Circulation. 2012;126:1717-1727.
19. Kovacic JC, Kini A, Banerjee S, et al. Patients with 3-vessel coronary artery disease and impaired ventricular function undergoing PCI with Impella 2.5 hemodynamic support have improved 90-day outcomes compared to intra-aortic balloon pump: a sub-study of the PROTECT II trial. J Interv Cardiol. 2015;28:32-40.
20. Tomasello SD, Boukhris M, Ganyukov V, et al. Outcome of extracorporeal membrane oxygenation support for complex high-risk elective percutaneous coronary interventions: a single-center experience. Heart Lung. 2015;44:309-313.
Sahil Khera, MD, MPH
Massachusetts General Hospital
Harvard Medical School
Boston, Massachusetts
Disclosures: None.
Srihari S. Naidu, MD
Westchester Medical Center
at New York Medical College
Valhalla, New York
srihari.naidu@wmchealth.org
Disclosures: None.
Advertisement
Advertisement

