Advertisement
Advertisement
January/February 2018
Impact of Radial Access on Coronary Intervention Outcomes
Coronary intervention outcomes can be improved with radial access—time for radial first.
Figure 1. Radial artery anatomy. Reprinted from Wikimedia Commons, the free media repository. Gray’s Anatomy plates, plate 528. https://commons.wikimedia.org. Accessed December 5, 2017.
Radial artery access for diagnostic coronary angiography was first reported by Lucien Campeau in 1989, followed by successful percutaneous coronary intervention (PCI) using Palmaz-Schatz stents through radial access (RA) in 1993.1,2 In his initial study, Campeau successfully achieved access in 88 of 100 patients and selectively engaged the coronary arteries without encountering difficulty in catheter manipulation or significant arterial spasm. The potential to decrease vascular complications by accessing an artery where the anatomy allows for easier compression and hemostasis (thus limiting access site bleeding and minimizing blood loss in the event of a complication and/or use of anticoagulants) and affording greater comfort to the patient was undoubtedly appealing (Figure 1).3
Despite this, RA did not gain popularity for coronary angiography and PCI until recent years, likely because sheaths and catheters were too large for radial insertion during the early days of coronary intervention. As multiple studies demonstrated that periprocedural bleeding—most often related to femoral access (FA)—is associated with an increased risk of major adverse cardiovascular events (MACEs),4,5 early adopters of RA began touting the safety of the approach versus FA.
Over the past few decades, multiple observational studies and randomized trials have evaluated the safety of RA and FA approaches, both in the setting of acute coronary syndromes (including ST-segment elevation myocardial infarction [STEMI]) and for elective procedures. Data supporting a benefit for RA versus FA with regard to decreased bleeding and vascular complications, and especially mortality, were initially met with some skepticism.6-9 Concerns regarding the feasibility of completing the RA procedure using catheters designed for FA, postprocedural radial artery occlusion, and recognition of a procedural learning curve were some of the reasons why many physicians resisted the RA strategy.10 Despite data continuing to support RA over FA, operators in the United States have been slower to adopt RA in comparison to Europe and Japan, where the proportion of RA cases approaches 50%.11
According to data from the National Cardiovascular Data Registry (NCDR) CathPCI Registry, RA rates in the United States have risen from 1.32% of cases in 2004 to 2007 to approximately 16% in 2008 to 2011.12 The availability of ultrasound guidance, specialized micropuncture and angiocatheter kits, low-profile introducer sheaths for smaller radial vessels, hydrophilic guidewires, and simple and effective compression devices have made RA more straightforward as compared to just a decade ago (Figure 2). Aside from radial artery loops, excessive tortuosity, and/or significant arterial spasm, the radial artery anatomy has proven to be favorable for vascular access angiography and PCI. Usage of an artery that supplies a distal limb with dual arterial supply and easy compressibility has an obvious appeal, and data comparing RA to FA have borne out the safety benefits.6-9
DATA HIGHLIGHTS
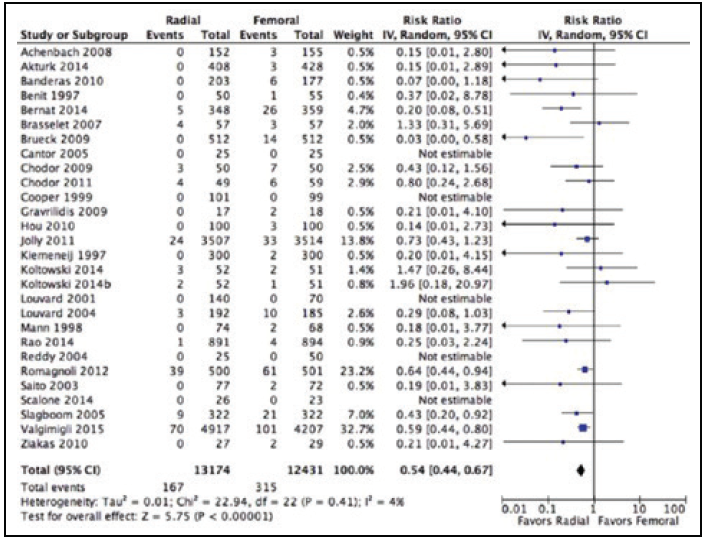
Figure 3. Forrest plot for the comparison of RA versus FA for major bleeding as outcome, defined according to the OASIS investigators. Reprinted with permission from Brener MI, Bush A, Miller JM, Hasan RK. Influence of radial versus femoral access site on coronary angiography and intervention outcomes: a systematic review and meta-analysis. Catheter Cardiovasc Interv. 2017;90:1093–1104.
In a review of the literature comparing RA versus FA from the 1980s to present day, conclusions range from no differences between the two techniques to marked reductions in postprocedural bleeding and lower mortality with the RA technique.6-9 Although evidence that RA reduces rates of bleeding has remained fairly consistent across studies over time, more data from larger randomized trials also suggest a potential decrease in the risk of mortality (Figure 3). The factors paramount to interpretation of the literature comparing RA to FA, beyond statistical power, include definition of bleeding events and operator/center experience with RA.
The RIVAL trial published in 2011 enrolled 7,021 patients with acute coronary syndrome who were randomized to RA versus FA.6 RIVAL was a randomized, parallel-group, multicenter effort. The primary outcome was a composite of death, myocardial infarction (MI), stroke, or noncoronary artery bypass graft (CABG)-related major bleeding at 30 days with a secondary outcome of death, MI, or stroke at 30 days. Although the rates of the primary outcomes and major non–CABG-related bleeding were similar in both access strategies, there was a significantly lower incidence of major vascular complications at 30 days in the RA group. This was defined by a decrease of 1.2% in the rate of large hematoma in the radial group compared with 3% in the femoral group (hazard ratio [HR], 0.40; 95% confidence interval [CI], 0.28–0.57; P < .0001), as well as a decrease of 0.2% versus 0.6% for pseudoaneurysm requiring closure, respectively (HR, 0.30; 95% CI, 0.13–0.71; P = .006). The caveats to these results is that the major non–CABG-related bleeding definition in the RIVAL trial was relatively conservative. For instance, utilizing the definition of non–CABG-related major bleeding and major vascular complications from the ACUITY trial,13 the access site–related bleeding rate was significantly lower with RA in comparison to FA (1.9% vs 4.5%; HR, 0.43; 95% CI, 0.32–0.57; P < .0001). In addition, examining the primary outcome in the study as assessed by centers with the highest radial PCI volume, there was a statistically significant decrease of 1.6% in the RA arm versus 3.2% in the FA arm (HR, 0.49; 95% CI, 0.28–0.87; P = .015). A similar benefit of RA versus FA was also observed with regard to the secondary endpoint of death, MI, or stroke (1.3% vs 2.7%; HR, 0.50; 95% CI, 0.27–0.92; P = .027), as well as for the major vascular complication rate.
The RIFLE-STEACS trial published in 2012, also a multicenter, randomized, and parallel-group study, enrolled only STEMI patients.7 The size of the trial (N = 1,001) was significantly smaller than that of RIVAL. The primary endpoint was the 30-day rate of net adverse clinical events (NACEs), defined as a composite of cardiac death, stroke, MI, target lesion revascularization, and bleeding, and individual components of NACEs and length of hospital stay were secondary endpoints. The primary endpoint was lower in the RA arm (13.6%) versus the FA arm (21%) (95% CI, 2.7%–12%; P = .003). The secondary endpoint of shorter coronary care unit stay was also noted in the RA arm at 3 days (range, 2–4 days) versus 4 days (range, 3–5 days) in the FA arm (P < .001). Bleeding, defined as any overt and actionable non–CABG-related hemorrhage with a ≥ 3 g/dL decrease in hemoglobin requiring prompt evaluation by a provider and leading to an increased level of care, was also lower at 7.8% versus 12.2% (95% CI, 2.7%–12%; P = .026).
A notable difference compared to RIVAL is that RA was also associated with a lower cardiac mortality of 5.2% versus 9.2% (95% CI, 0.8–7.3%; P = .020). Essentially, RA had a 4% absolute reduction in 30-day mortality and a 4.2% absolute reduction in access site bleeding, leaving no difference in nonaccess site bleeding (53% of all bleeding events), as one would suspect. Unfortunately, much of the cardiac mortality was within 48 hours of the STEMI, and it was challenging to determine the role that bleeding played in these deaths. Importantly, in this STEMI-only trial, door-to-balloon time was not significantly prolonged in the RA arm and no difference in angiographic failure rates was observed. These findings provided a measure of reassurance to those operators weary of RA under the circumstances of STEMI.
Another STEMI-only trial of RA versus FA was the STEMI-RADIAL study published in 2014, which randomized 704 patients to FA versus RA with a primary endpoint of cumulative incidence of major bleeding and vascular access site complications at 30 days and NACE as a secondary endpoint, along with access site crossover, contrast volume, duration of intensive care unit (ICU) stay, and death at 6 months.9 The definition of major bleeding adopted for STEMI-RADIAL was the HORIZONS-AMI criteria,14 with only hematomas > 15 cm recorded so that smaller hematomas would not affect event rates. This trial also demonstrated significantly lower incidences of major bleeding and access site complications in the RA group, with the primary endpoint occurring in 1.4% of patients as compared with 7.2% in the FA group (P = .0001). The NACE rate was also lower at 4.6% versus 11% (P = .0028), respectively. Although mortality was no different at 30 days and 6 months, contrast volume and duration of stay in the ICU was also significantly decreased with the radial approach.
The mechanisms by which access site choice could affect mortality have not been unequivocally elucidated and remain a matter of debate. Significant bleeding in the FA patient can cause hemodynamic compromise and a potentially fatal outcome if not expeditiously remedied. In addition, such a bleeding event can itself not only activate a systemic inflammatory cascade but also the coagulation cascade, which can potentially pose a risk to the patient with an acute coronary syndrome in which the treatments employed are attempting to combat these very responses. Moreover, a bleeding complication often necessitates at least temporary cessation of antiplatelet and antithrombotic therapies, potentially increasing the risk for further atherothrombotic events. Furthermore, bleeding requiring transfusion is accompanied by risks inherent with a blood transfusion, such as volume load and transfusion reactions.
The most recent systematic review and meta-analysis of the randomized trial data comparing RA to FA showed that the primary composite MACE endpoint was lower in RA, and this was driven by a reduction in all-cause mortality.15 Still, controversy and skepticism exist as to whether RA truly can decrease mortality. In contrast, current data are unequivocal in showing that bleeding and vascular complication rates are lower with RA compared to FA, and these findings have generally gained widespread recognition and acceptance.16
Another important point raised by the RIFLE-STEACS, STEMI-RADIAL, and other trials is the significantly decreased length of hospital/ICU stay. As pressure mounts on the United States health care system to reduce costs, interventions (both medical and procedural) will need to evolve to deliver quality care at lower financial costs. If RA decreases complication rates and expedites safe discharge of the patient in a shorter time frame, the reduction in costs has the potential to be significant. A recent study by Amin et al analyzing the NCDR CathPCI Registry linked to Medicare claims of patients undergoing PCI (excluding primary PCI), both on an inpatient and outpatient basis, demonstrated that, by shifting practice from RA use in 9% of patients to approximately 30% to facilitate quicker discharge, a hospital performing 1,000 elective PCI procedures per year could reduce costs by approximately $1 million annually.17
CONSIDERATIONS FOR RADIAL ACCESS
Although the data comparing RA to FA can seem overwhelming at times, often the decision by the operator can boil down to a number of situation-specific factors. First, patient preference can often influence operator decision. The data support that patients prefer RA over FA due to comfort and quicker ambulation and discharge times. While catheter manipulation and subsequent successful angiography of the coronary arteries via RA can be achieved the majority of the time, there are instances in which the operator may be unable to successfully cannulate the radial artery, engage a vessel with a catheter, and/or obtain appropriate guide catheter backup support to perform PCI and therefore would require FA. Although there was significant statistical heterogeneity, the recent meta-analysis by Brener et al showed that RA was associated with a higher rate of procedural failure.15 For example, the RIFLE-STEACS trial showed no difference in angiographic failure rate in FA versus RA; however, the crossover rate in the STEMI-RADIAL trial was 3.7% in the RA arm versus 0.6% in the FA arm (P = .0034).7,9
Patients with chronic kidney disease also present challenges; for example, when the operator must consider using an alternative to RA in those with existing arteriovenous fistulas or being mindful of preserving arterial vasculature for potential creation of future dialysis access. A history of Raynaud phenomenon is also widely cited as a relative contraindication to RA. In patients with severely calcified common femoral arteries and extensive peripheral artery disease, FA can be a challenge as well as present an increased risk for vascular complication, pushing an operator toward RA.
Last, fluoroscopy times with RA compared to FA have been another point of contention among operators analyzing the benefits of the RA approach. For instance, the RIVAL trial showed, on average, fluoroscopy time was 1.3 minutes longer in the RA arm (P < .0001). However, the median radiation dose measured by air kerma was only higher by a nominal measurement of 1,046 mGy (range, 584–1,591 mGy) for RA versus 930 mGy (range, 538–1,572 mGy) for FA (P = .051) overall. These differences were present only for the lower-volume centers/operators, whereas high-volume centers had the lowest air kerma dose regardless of access site approach.18 Aside from strong patient preference or anatomic/medical contraindication to the use of RA, the experience of the center/operators significantly impacts the successful completion of RA, consistent with the learning curve for this approach.
Cardiology training programs should employ a radial-first approach in light of the available data; however, there is still value in learning to safely obtain FA in the appropriate situations. Apart from when RA is not feasible and urgent FA is required in time-sensitive situations, with mechanical circulatory support becoming more widespread for the purpose of assisting high-risk PCIs or as a bridging therapy in the unstable patient in cardiogenic shock, adeptness in FA is a requirement. Skill in FA and management of FA site complications is of utmost importance in patients in whom larger-bore vascular access is necessary, including mechanical circulatory support and structural heart disease interventions. However, the preponderance of RA at some institutions may limit the training experience with FA, resulting in a challenging situation in which new operators may be less facile with FA and femoral hemostasis as a result. Nonetheless, current data and experience still favor a radial-first strategy, even if one is skeptical of the impact of access site choice on mortality.
CONCLUSION
The data on RA compared to FA for coronary angiography over the past few decades have been varied with regard to an effect on morality; however, the decreases reported in vascular complications and bleeding rates are fairly robust and consistent. Over this time, operators in the United States have been slower to adopt the RA approach compared to those in Europe and Asia. There are sufficient data at this point to confirm the benefits of RA with regard to postprocedural complications, comfort, and health care costs. Interventional cardiologists should incorporate a radial-first approach in their practice, with the caveat that the femoral approach may be favored in specific instances and procedures.
1. Campeau L. Percutaneous radial artery approach for coronary angiography. Cathet Cardiovasc Diagn. 1989;16:3-7.
2. Kiemeneij F, Laarman GJ. Percutaneous transradial artery approach for coronary stent implantation. Cathet Cardiovasc Diagn. 1993;30:173-178.
3. Cooper CJ, El-Shiekh RA, Cohen DJ. Effect of transradial access on quality of life and cost of cardiac catheterization: a randomized comparison. Am Heart J. 1999;138:430-436.
4. Doyle BJ, Rihal CS, Gastineau DA, et al. Bleeding, blood transfusion, and increased mortality after percutaneous coronary intervention: implications for contemporary practice. J Am Coll Cardiol. 2009;53:2019-2027.
5. Rao SV, Dai D, Subherwal S, et al. Association between periprocedural bleeding and long-term outcomes following percutaneous coronary intervention in older patients. JACC Cardiovasc Interv. 2012;5:958-965.
6. Jolly SS, Yusuf S, Cairns J, et al. Radial versus femoral access for coronary angiography and intervention in patients with acute coronary syndromes (RIVAL): a randomised, parallel group, multicentre trial. Lancet. 2011;377:1409-1420.
7. Romagnoli E, Biondi-Zoccai G, Sciahbasi A, et al. Radial versus femoral randomized investigation in ST-segment elevation acute coronary syndrome: the RIFLE-STEACS (Radial Versus Femoral Randomized Investigation in ST-Elevation Acute Coronary Syndrome) study. J Am Coll Cardiol. 2012;60:2481-2489.
8. Valgimigli M, Gagnor A, Calabró P, et al. Radial versus femoral access in patients with acute coronary syndromes undergoing invasive management: a randomised multicentre trial. Lancet. 2015;385:2465-2476.
9. Bernat I, Horak D, Stasek J, et al. ST-segment elevation myocardial infarction treated by radial or femoral approach in a multicenter randomized clinical trial: the STEMI-RADIAL trial. J Am Coll Cardiol. 2014;63:964-972.
10. Rao SV, Cohen MG, Kandzari DE, et al. The transradial approach to percutaneous coronary intervention: historical perspective, current concepts, and future directions. J Am Coll Cardiol. 2010;55:2187-2195.
11. Caputo RP, Tremmel JA, Rao S, et al. Transradial arterial access for coronary and peripheral procedures: Executive summary by the transradial committee of the SCAI. Catheter Cardiovasc Interv. 2011;78:823-839.
12. Feldman DN, Swaminathan RV, Kaltenbach LA, et al. Adoption of radial access and comparison of outcomes to femoral access in percutaneous coronary intervention: an updated report from the national cardiovascular data registry (2007-2012). Circulation. 2013;127:2295-2306.
13. Stone GW, McLaurin BT, Cox DA, et al. Bivalirudin for patients with acute coronary syndromes. N Engl J Med. 2006;355:2203-2216.
14. Stone GW, Witzenbichler B, Guagliumi G, et al. Bivalirudin during primary PCI in acute myocardial infarction. N Engl J Med. 2008;358:2218-2230.
15. Brener MI, Bush A, Miller JM, Hasan RK. Influence of radial versus femoral access site on coronary angiography and intervention outcomes: a systematic review and meta-analysis. Catheter Cardiovasc Interv. 2017;90:1093-1104.
16. Ferrante G, Rao SV, Jüni P, et al. Radial versus femoral access for coronary interventions across the entire spectrum of patients with coronary artery disease: a meta-analysis of randomized trials. JACC Cardiovasc Interv. 2016;9:1419-1434.
17. Amin AP, Patterson M, House JA, et al. Costs associated with access site and same-day discharge among Medicare beneficiaries undergoing percutaneous coronary intervention: an evaluation of the current percutaneous coronary intervention care pathways in the United States. JACC Cardiovasc Interv. 2017;10:342-351.
18. Jolly SS, Cairns J, Niemela K, et al. Effect of radial versus femoral access on radiation dose and the importance of procedural volume: a substudy of the multicenter randomized RIVAL trial. JACC Cardiovasc Interv. 2013;6:258-266.
M. Imran Aslam, MD
Division of Cardiology
Johns Hopkins University School of Medicine
Baltimore, Maryland
Disclosures: None.
Rani K. Hasan, MD, MHS
Division of Cardiology
Johns Hopkins University School of Medicine
Baltimore, Maryland
(410) 614-1132; rani.hasan@jhmi.edu
Disclosures: None.
Advertisement
Advertisement

