Advertisement
Advertisement
September/October 2016
Managing the Risk of Coronary Occlusion During TAVR
Effective planning for valve retrieval and coronary protection is essential.
By Carlos E. Sanchez, MD, FACC, FSCAI; Arash Arshi, MD, FACC, FSCAI; and Steven J. Yakubov, MD, FACC, FSCAI
Transcatheter aortic valve replacement (TAVR) is a well-established therapy that has revolutionized the management of patients with severe aortic stenosis who are deemed suboptimal candidates for surgery.1-3 Despite increasing experience with and understanding of the procedure and patient selection, TAVR is still associated with unexpected complications, such as paravalvular aortic regurgitation, conduction disturbances, cardiac perforation, and ostial coronary occlusion.
Obstruction of the coronary ostium during a native valve TAVR is relatively infrequent, occurring in < 1% of cases. Left main coronary occlusion is more commonly involved, whereas right coronary artery occlusion rarely happens after valve deployment.4 However, this complication can result in acute hemodynamic collapse and is associated with a high mortality rate.4,5 Therefore, rapid recognition, institution of hemodynamic support, and reestablishment of coronary flow are critical for patient survival.
CLINICAL PRESENTATION
Persistent severe hypotension after TAVR is the most common presentation for ostial coronary occlusion due to significant myocardial ischemia. Onset of symptoms usually occurs immediately after valve implantation in 88% of patients; however, late presentation has also been described within the first 48 hours after valve implantation. Approximately 50% of patients present with ST-segment changes determined by electrocardiography, and 25% present with ventricular arrhythmias during the procedure.4,5 Therefore, the presence of persistent procedural hypotension after valve implantation, even without clear electrocardiographic ST-segment changes, should prompt immediate echocardiography and/or coronary angiography to evaluate wall motion abnormalities and identify coronary obstruction, respectively.
MECHANISM OF CORONARY OBSTRUCTION
Understanding the mechanisms involved in coronary obstruction is important in order to risk stratify patients during preprocedure planning for TAVR. The most common mechanism of coronary artery obstruction after TAVR is displacement of the native calcified valve leaflet over the coronary ostium, described as the main cause in 98% of patients.4,5 Other possible mechanisms for coronary obstruction include impingement of the coronary ostium by the transcatheter valve scaffold or leaflets.4,5
Although the mechanisms that lead to coronary obstruction have been very well described, the clinical risk factors that influence its occurrence are less clear. Multiple important anatomic risk factors have been associated with coronary occlusion. A low takeoff of the coronary artery in relation to the aortic annulus has been strongly associated as one of the most important risk factors contributing to this complication. In fact, professional societies advise that a distance ≤ 10 mm between the coronary ostium takeoff to the nadir of the corresponding sinus increases the risk of coronary occlusion during TAVR.6 Recent data have shown that the mean height of the left main coronary ostium in reported cases of coronary obstruction was between 10 and 11 mm.4,5 However, > 25% of coronary obstruction cases had a left main coronary height of > 12 mm, suggesting that additional risk factors may also play a role in the occurrence of this complication.5 Effacement of the aortic root with sinuses of Valsalva measuring < 30 mm in diameter and a narrow sinotubular junction have also been described as potential risk factors for coronary occlusion after TAVR.4,5 Finally, coronary obstruction has been associated with a three- to fourfold increased risk after valve-in-valve TAVR when compared with native valve TAVR5,7 with an incidence described between 2.5% to 3.5%.7,8
Multidetector CT is an essential tool in the workup of patients who are being considered for TAVR. Important anatomic structures and measurements include the height of the coronary ostia in relation to the aortic valve annulus, the width and height of the sinus of Valsalva, and the width of the sinotubular junction.9
MANAGEMENT OF CORONARY OBSTRUCTION
When the risk of coronary obstruction is high, additional safety measures should be implemented during the TAVR procedure. Some operators recommend performing contrast aortography during balloon aortic valvuloplasty to assess the patency of the coronary arteries.4 When multiple anatomic parameters suggest potential coronary artery compromise, coronary protection can be performed with a guidewire positioned in the coronary artery and left in place during valve deployment. Some operators advocate the use of an undeployed stent distal to the coronary ostium ready to be pulled back and deployed if necessary.7 Finally, the use of a transcatheter valve that can be retrieved or repositioned after valve implantation, such as the CoreValve Evolut R (Medtronic) or Lotus (Boston Scientific Corporation), may be preferred in high-risk cases.5,7
Once the valve is deployed and coronary obstruction has occurred, the measures required to resolve this issue depend on the valve used. Obstruction with use of the CoreValve device can be resolved with snaring one or both tabs of the frame and pulling the deployed valve above the sinotubular junction. Ideally, both tabs are pulled with careful attention to the risk of aortic dissection. One tab is usually accessed from the femoral approach, whereas the other is accessed through the radial approach. When a new valve is being reinserted, holding onto the retrieved valve is essential to avoid forward migration. Obstruction after deployment of any Sapien valve (Edwards Lifesciences) requires immediate cannulation of the affected coronary artery with a guide catheter for balloon angioplasty of the ostium and often coronary stent deployment. It is necessary to use a high-pressure balloon after stent deployment to obviate the effect of ostial deformation.
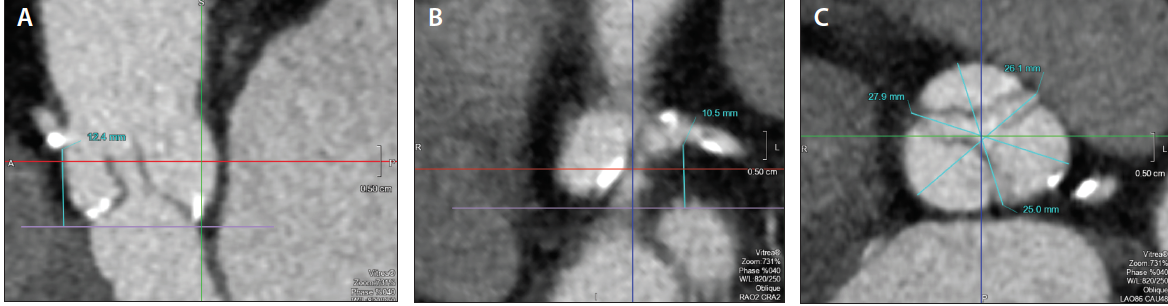
Figure 1. CT angiographic measurements were obtained, the results of which were notable for low coronary heights from the aortic annulus to the lower margin of the ostia of the right (A) and left main (B) coronary arteries. The diameters of the sinuses of Valsalva were noted to be narrow (C), whereas the aortic annulus perimeter measured 70 mm, within the range of a 26-mm CoreValve Evolut R device.
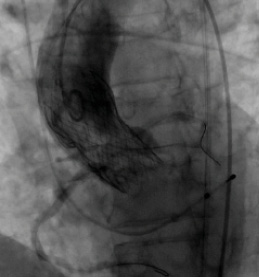
Figure 2. After deployment of the CoreValve Evolut R device, the coronary guide catheters were withdrawn while maintaining wire access to the coronary arteries. Aortography was performed, demonstrating patency of the coronary ostia.
In the case of a patient with low takeoffs of the coronary ostia and narrow sinuses of Valsalva (Figure 1), the preference at our institution is to protect the potentially affected coronary arteries with coronary guidewires prior to placement of the valve. We withdraw the guide catheters above the level of the leaflet insertion of the prosthetic valve, leaving the guidewire in place, so that patency of the coronary ostia can be clearly visualized. After deployment of the transcatheter valve, aortography is performed to demonstrate patency of the coronary arteries (Figure 2).
Case Example
A particularly high-risk scenario for coronary occlusion arises in the setting of valve-in-valve TAVR. For example, a patient had undergone a recent TAVR with a 29-mm CoreValve device, which migrated during final release just above the aortic annulus. Initially, there was mild to moderate paravalvular regurgitation with acceptable hemodynamic parameters. Over the subsequent week, the patient developed progressive congestive heart failure and was found to have severe paravalvular regurgitation. We determined that valve in valve would be the best option. The preferred initial strategy would be to reposition the initial CoreValve higher in the ascending aorta, which could be accomplished by snaring the valve at one or both posts and mechanically pulling the valve. Additionally, balloon inflation within the valve may be helpful in repositioning the valve. However, due to severe aortic atherosclerosis and calcification, the balloon inflation maneuver would carry a high risk of aortic dissection and atheroembolism in our patient. We instead opted to insert a second 29-mm CoreValve device while protecting the native left main coronary artery with a guidewire and stent. Upon passage of the second CoreValve delivery system across the annulus, the patient developed hemodynamic compromise and new left ventricular dysfunction, presumably secondary to obstruction of blood flow to the left main coronary ostium by the fixed leaflets of the initial prosthetic valve.
The supra-annular location of the CoreValve device may cause obstruction above the level of the sinotubular junction due to the long prosthetic leaflets fixed in the open position along with the inner skirt, essentially creating a tube extending from the annulus above the level of the sinotubular junction. The patient was prophylactically placed on cardiopulmonary bypass. We protected the left main coronary artery with a coronary guidewire, and an undeployed drug-eluting stent was temporarily positioned in the left coronary system.
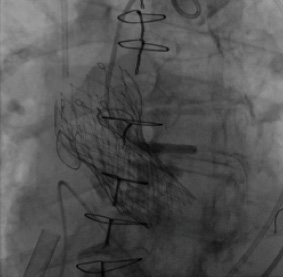
Figure 3. The “snorkel” technique provides left main coronary blood flow. A 3- X 30-mm Resolute drug-eluting stent (Medtronic) was positioned, extending from above the prosthetic valve leaflet insertion line into the left main coronary artery.
After successful release of the second CoreValve device at the level of the aortic annulus, the stent was repositioned and deployed in the native left main artery, with its proximal edge extending above the level of the initial CoreValve leaflet insertion line (Figure 3). This “snorkel” technique allowed blood flow into the left coronary system, enabling the patient to be weaned immediately from cardiopulmonary support.
CONCLUSION
To obviate the chance of coronary obstruction, it is essential to have an effective preprocedural plan in place and to effectively use anatomic information from CT and assess valve leaflet characteristics from echocardiography. Immediate recognition of obstruction through echocardiography and angiography is of paramount importance. A plan for resolution of coronary obstruction, including valve retrieval and the institution of support measures and coronary intervention, should be part of every TAVR evaluation.
1. Nishimura RA, Otto CM, Bonow RO, et al. 2014 AHA/ACC guideline for the management of patients with valvular heart disease: executive summary: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines. J Am Coll Cardiol. 2014;63:e57-185.
2. Leon MB, Smith CR, Mack M, et al. Transcatheter aortic-valve implantation for aortic stenosis in patients who cannot undergo surgery. N Engl J Med. 2010;363:1597-1607.
3. Adams DH, Popma JJ, Reardon MJ, et al. Transcatheter aortic-valve replacement with a self-expanding prosthesis. N Engl J Med. 2014;370:1790-1798.
4. Ribeiro HB, Nombela-Franco L, Urena M, et al. Coronary obstruction following transcatheter aortic valve implantation: a systematic review. JACC Cardiovasc Interv. 2013;6:452-461.
5. Ribeiro HB, Webb JG, Makkar RR, et al. Predictive factors, management, and clinical outcomes of coronary obstruction following transcatheter aortic valve implantation: insights from a large multicenter registry. J Am Coll Cardiol. 2013;62:1552-1562.
6. Holmes DR Jr, Mack MJ, Kaul S, et al. ACCF/AATS/SCAI/STS expert consensus document on transcatheter aortic valve replacement. J Am Coll Cardiol. 2012;59:1200-1254.
7. Dvir D, Leipsic J, Blanke P, et al. Coronary obstruction in transcatheter aortic valve-in-valve implantation: preprocedural evaluation, device selection, protection, and treatment. Circ Cardiovasc Interv. 2015;8:e002079.
8. Dvir D, Webb J, Brecker S, et al. Transcatheter aortic valve replacement for degenerative bioprosthetic surgical valves: results from the global valve-in-valve registry. Circulation. 2012;126:2335-2344.
9. Achenbach S, Delgado V, Hausleiter J, et al. SCCT expert consensus document on computed tomography imaging before transcatheter aortic valve implantation (TAVI)/transcatheter aortic valve replacement (TAVR). J Cardiovasc Comput Tomogr. 2012;6:366-380.
Carlos E. Sanchez, MD, FACC, FSCAI
OhioHealth Research Institute
Riverside Methodist Hospital
Columbus, Ohio
Disclosures: None.
Arash Arshi, MD, FACC, FSCAI
OhioHealth Research Institute
Riverside Methodist Hospital
Columbus, Ohio
Disclosures: None.
Steven J. Yakubov, MD, FACC, FSCAI
OhioHealth Research Institute
Riverside Methodist Hospital
Columbus, Ohio
steven.yakubov@gmail.com
Disclosures: Steering committee for CoreValve clinical trials; proctor and medical advisory board, Medtronic.
Advertisement
Advertisement
