Advertisement
Advertisement
July/August 2024
Essentials of Coronary Calcium Evaluation
Role of invasive and noninvasive imaging in assessing coronary calcium.
By Sarah Malik, MD; Evan Shlofmitz, DO; and Richard A. Shlofmitz, MD
Percutaneous coronary intervention (PCI) of calcified lesions is associated with stent underexpansion, which increases the risk of acute stent thrombosis and in-stent restenosis.1-4 In this regard, assessment of coronary artery calcification (CAC) plays a critical role in PCI planning and optimization. Intravascular imaging (IVI), such as intravascular ultrasound (IVUS) or optical coherence tomography (OCT), is an invasive intraprocedural technique that allows the visualization of CAC burden and morphology.5-7 Calcium scoring systems based on IVI findings have been developed to predict the likelihood of stent underexpansion and identify the calcified lesions that will benefit from advanced plaque modification prior to stent implantation. Coronary CTA (CCTA) is a noninvasive imaging tool that can also be used to visualize and characterize calcium morphology in its longitudinal and circumferential extension.8 Compared with IVI, CCTA can provide detailed information on CAC prior to the procedure, which may help operators and patients prepare for the complex procedure in advance, thereby increasing procedural safety and efficacy. This article discusses the role of IVI and CCTA in assessing CAC to strategize and optimize PCI.
IVI AND PCI FOR CALCIFIED LESIONS
CAC continues to pose one of the greatest challenges during PCI. Although fluoroscopy can detect moderate to severe CAC, it has limited accuracy and lacks the capability to evaluate detailed features and burden of CAC.9,10 Compared with angiography, IVI not only increases diagnostic accuracy of CAC but also enables the assessment of morphologic features of CAC that are associated with suboptimal PCI outcomes.9 Thus, IVI has been used to identify calcified lesions that may require advanced calcium modification therapies. In addition, IVI can be used to optimize stent implantation by evaluating appropriate landing zones, stent size and length, stent expansion and minimal stent area, edge dissection, and untreated inflow/outflow residual disease. Consequently, multiple randomized trials and a recent meta-analysis demonstrated clear benefit of IVI use in the treatment of heavily calcified coronary lesions.11
IVUS
CAC appears as a highly echogenic area with acoustic shadowing in IVUS (Figure 1A). Morphologic features of CAC seen in IVUS that are associated with stent underexpansion include (1) calcium angle > 270° for > 5 mm, (2) circumferential calcium (360°), (3) calcified nodule, and (4) small vessel diameter < 3.5 mm (Figure 1A).12 A study by Zhang et al suggested that up-front advanced calcium modification therapies would be required for calcified lesions with two or more of these features to achieve better stent expansion.12
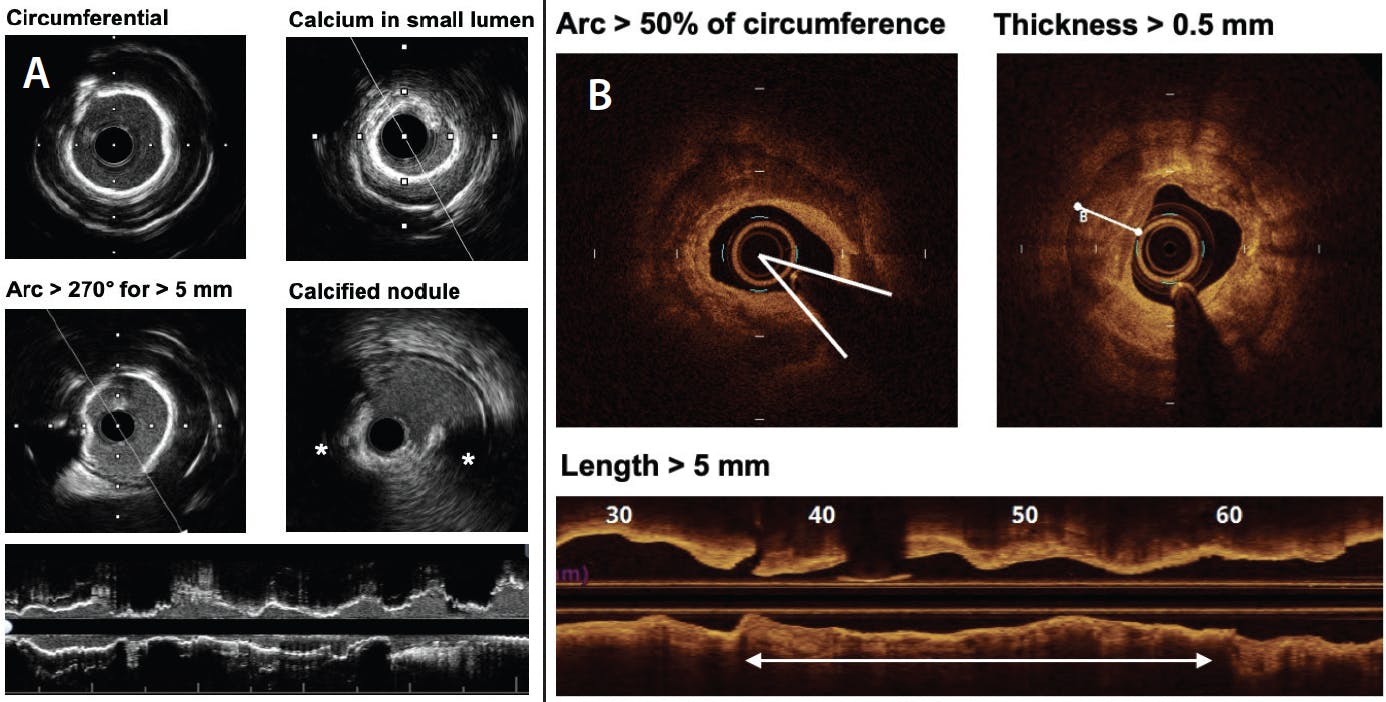
Figure 1. IVUS and OCT calcium assessment. Components of calcium scoring systems by IVUS (A) and OCT (B). In IVUS, calcium appears as a highly echogenic area with acoustic shadowing. Thus, calcium thickness cannot be assessed by IVUS. The IVUS calcium scoring system includes calcium arc, presence of calcified nodule, and small vessel diameter. In contrast, OCT can provide information on calcium thickness. The OCT calcium scoring system is derived from calcium arc, length, and thickness.
OCT
In OCT, CAC can be visualized as a clearly delineated, low-attenuating area (Figure 1B). Compared with IVUS, OCT allows more detailed evaluation of CAC because calcium arc as well as calcium thickness can be assessed by OCT. Therefore, OCT enables volumetric assessment of CAC by measuring calcium thickness, arc, and length, all of which are included in the OCT-based calcium scoring system. A study by Fujino et al suggested that PCI of calcified lesions with maximum calcium arc > 180°, maximum calcium thickness > 0.5 mm, and calcium length > 5 mm would result in suboptimal stent expansion, thereby benefitting from up-front advanced calcium modification therapies (rule of “5”: arc > 50% of circumference, thickness > 0.5 mm, and length > 5 mm) (Figure 1B).13
CORONARY CALCIUM ASSESSMENT BY CCTA FOR PCI PLANNING
Similar to IVI, CCTA can provide detailed information on plaque characteristics, including CAC (Figure 2). A study by Monizzi et al suggested that CCTA-derived calcium arc, thickness, and length are reliable using OCT as a reference, although volumetric measurement of CAC by CCTA can be overestimated by 60%.8,14,15 Therefore, CCTA can provide a unique opportunity to evaluate characteristics of CAC associated with suboptimal procedural results even before bringing the patient to the catheterization laboratory.15 For example, a per-lesion calcium score ≥ 453 and calcification arc ≥ 270° on CCTA can suggest the need for rotational atherectomy.14 One of the CCTA features that cannot be assessed by IVI is calcium density measured in Hounsfield units (HU), which reflects resilience and resistance to fracture.15 Calcified lesions with higher HU are less likely to be fractured during PCI than those with lower HU (Figure 2A) and thus may benefit from up-front advanced calcium modification therapies.15,16 Last but not least, CCTA with three-dimensional (3D) reconstruction can help operators understand calcium distribution in longitudinal and circumferential extension and location in relation to vessel circumference and predict wire position and bias (Figure 2B and 2C).15 This information can help not only guide PCI but also choose the appropriate advanced calcium modification therapy, especially because unfavorable wire bias will reduce the efficacy of atherectomy while increasing the risk of complications.
BENEFITS OF UP-FRONT CALCIUM ASSESSMENT BY CCTA AND FUTURE DIRECTIONS
Although CCTA can provide comprehensive information on CAC for pre-PCI planning, there is no CCTA-dedicated calcium scoring system like IVI, which warrants further investigation. Nevertheless, up-front calcium assessment by CCTA could provide unique benefits for PCI in many ways. First, it can enhance procedural precision. CCTA provides not only comprehensive information on the entire coronary artery and CAC but also plaque vulnerability, physiologic significance and disease pattern, and subtended myocardial mass. Therefore, operators will have enough time to plan for the best possible PCI strategy using all the information together. Second, procedural efficacy can be maximized. Based on calcium morphology, severity, and density from CCTA, operators would already know which devices and modalities they need, thus minimizing procedural time and optimizing resource allocation. Third, it enables operators to discuss the complexity of the procedure, potential advanced techniques and equipment, and risks and benefits with the patient beforehand, ensuring thorough preparation and informed consent. This novel concept of CCTA-guided PCI, including dedicated assessment of CAC, will be tested in the ongoing P4 trial (NCT05253677). Nevertheless, CCTA cannot replace IVI during PCI, especially for optimization of stent placement. Eventually, CCTA and IVI will be used as complementary modalities to enhance precision, efficacy, and safety of PCI and improve patient outcomes.
CONCLUSION
Because angiography alone is limited in evaluating CAC, IVI has been used to understand the adverse features of CAC that prevent optimal stent expansion. IVI calcium scoring systems are derived from these adverse features of CAC and have remained useful intraoperative tools to determine the need for advanced calcium modification strategies. With advancement of technology, CCTA is increasingly used and allows reliable up-front calcium assessment, which may have additional benefits to enhance the precision, efficacy, and safety of PCI. Future studies are warranted to understand the complementary role of CCTA in assessing CAC for PCI planning and enhancing patient outcomes.
1. Hoffmann R, Mintz GS, Popma JJ, et al. Treatment of calcified coronary lesions with Palmaz-Schatz stents. An intravascular ultrasound study. Eur Heart J. 1998;19:1224-1231. doi: 10.1053/euhj.1998.1028
2. Kobayashi Y, Okura H, Kume T, et al. Impact of target lesion coronary calcification on stent expansion. Circ J. 2014;78:2209-2214. doi: 10.1253/circj.cj-14-0108
3. Taniwaki M, Radu MD, Zaugg S, et al. Mechanisms of very late drug-eluting stent thrombosis assessed by optical coherence tomography. Circulation. 2016;133:650-660. doi: 10.1161/circulationaha.115.019071
4. Song HG, Kang SJ, Ahn JM, et al. Intravascular ultrasound assessment of optimal stent area to prevent in-stent restenosis after zotarolimus-, everolimus-, and sirolimus-eluting stent implantation. Catheter Cardiovasc Interv. 2014;83:873-878. doi: 10.1002/ccd.24560
5. Lee JM, Choi KH, Song YB, et al. Intravascular imaging-guided or angiography-guided complex PCI. N Engl J Med. 2023;388:1668-1679. doi: 10.1056/NEJMoa2216607
6. Li X, Ge Z, Kan J, et al. Intravascular ultrasound-guided versus angiography-guided percutaneous coronary intervention in acute coronary syndromes (IVUS-ACS): a two-stage, multicentre, randomised trial. Lancet. 2024;403:1855-1865. doi: 10.1016/s0140-6736(24)00282-4
7. Stone GW, Christiansen EH, Ali ZA, et al. Intravascular imaging-guided coronary drug-eluting stent implantation: an updated network meta-analysis. Lancet. 2024;403:824-837. doi: 10.1016/s0140-6736(23)02454-6
8. Monizzi G, Sonck J, Nagumo S, et al. Quantification of calcium burden by coronary CT angiography compared to optical coherence tomography. Int J Cardiovasc Imaging. 2020;36:2393-2402. doi: 10.1007/s10554-020-01839-z
9. Barbato E, Gallinoro E, Abdel-Wahab M, et al. Management strategies for heavily calcified coronary stenoses: an EAPCI clinical consensus statement in collaboration with the EURO4C-PCR group. Eur Heart J. 2023;44:4340-4356. doi: 10.1093/eurheartj/ehad342
10. Mintz GS, Popma JJ, Pichard AD, et al. Patterns of calcification in coronary artery disease. A statistical analysis of intravascular ultrasound and coronary angiography in 1155 lesions. Circulation. 1995;91:1959-1965. doi: 10.1161/01.cir.91.7.1959
11. Shin D, Hong D, Singh M, et al. Intravascular imaging-guided percutaneous coronary intervention for heavily calcified coronary lesions: a systematic review and meta-analysis. Int J Cardiovasc Imaging. Published online June 14, 2024. doi: 10.1007/s10554-024-03150-7
12. Zhang M, Matsumura M, Usui E, et al. Intravascular ultrasound-derived calcium score to predict stent expansion in severely calcified lesions. Circ Cardiovasc Interv. 2021;14:e010296. doi: 10.1161/circinterventions.120.010296
13. Fujino A, Mintz GS, Matsumura M, et al. A new optical coherence tomography-based calcium scoring system to predict stent underexpansion. EuroIntervention. 2018;13:e2182-e2189. doi: 10.4244/eij-d-17-00962
14. Sekimoto T, Akutsu Y, Hamazaki Y, et al. Regional calcified plaque score evaluated by multidetector computed tomography for predicting the addition of rotational atherectomy during percutaneous coronary intervention. J Cardiovasc Comput Tomogr. 2016;10:221-228. doi: 10.1016/j.jcct.2016.01.004
15. Tajima A, Bouisset F, Ohashi H, et al. Advanced CT imaging for the assessment of calcific coronary artery disease and PCI planning. J Soc Cardiovasc Angiogr Interv. 2024;3:101299. doi: 10.1016/j.jscai.2024.101299
16. Gupta NP, Ansari MS, Kesarvani P, et al. Role of computed tomography with no contrast medium enhancement in predicting the outcome of extracorporeal shock wave lithotripsy for urinary calculi. BJU Int. 2005;95:1285-1288. doi: 10.1111/j.1464-410X.2005.05520.x
Advertisement
Advertisement

