Advertisement
Advertisement
January/February 2022
BASILICA for Coronary Obstruction Prevention During TAVR: Procedural Technique, Data, and Future Challenges
A technical guide to performing solo and doppio BASILICA.
By Giorgio A. Medranda, MD; Jaffar M. Khan, BM BCh, PhD; Christopher Bruce, MB ChB; Patrick Gleason, MD; Joe Xie, MD; Adam B. Greenbaum, MD; Robert J. Lederman, MD; Vasilis C. Babaliaros, MD; and Toby Rogers, MD, PhD
Although rare, iatrogenic coronary artery obstruction is a devastating complication of transcatheter aortic valve replacement (TAVR) that is associated with high morbidity and mortality.1-4 Patients with previous bioprosthetic aortic valves are at higher risk.5 The BASILICA (bioprosthetic or native aortic scallop intentional laceration to prevent iatrogenic coronary artery obstruction during TAVR) technique uses transcatheter electrosurgery before TAVR to lacerate the offending aortic leaflet(s) and maintain coronary perfusion after TAVR.6 This article details the BASILICA procedural technique for both a single leaflet (“solo”) and double leaflet (“doppio”), clinical data, and future directions.
PROCEDURAL TECHNIQUE
Materials
Table 1 outlines the specific equipment that is required for performing both solo and doppio BASILICA.
Preprocedural Planning
Although detailed step-by-step preprocedural planning for BASILICA is beyond the scope of this article, it has been previously described in detail by Lederman et al.7
Step 1: Vascular Access
Common femoral arterial access is achieved using an ultrasound-guided micropuncture technique. Venous access for temporary pacing can be selected per institutional standard practice. Once all access sites have been successfully obtained, full-dose heparin is administered to achieve an activated clotting time > 250 seconds.
Solo BASILICA. This requires the same number and size of access sites as conventional transfemoral TAVR: one large-bore femoral arterial access (initially with a 14- to 18-F Gore DrySeal Flex [Gore & Associates]) is used for one BASILICA catheter and then to deliver the transcatheter heart valve (or to exchange for a dedicated TAVR sheath). A contralateral small-bore arterial access (eg, 6 F) is used for the second BASILICA catheter, followed by the pigtail catheter for TAVR.
Doppio BASILICA. The small-bore contralateral arterial access is replaced by a second large-bore arterial access (14- to 18-F DrySeal). Each large-bore sheath will accommodate both BASILICA catheters for one leaflet.
Step 2: Leaflet Traversal
First, the aortic valve is crossed in the usual fashion from the contralateral small-bore access to then introduce the left ventricular outflow tract (LVOT) catheter (Table 2). An 0.018-inch guidewire is then placed through the LVOT catheter into the left ventricle to anchor the system, allowing the LVOT catheter to be easily advanced back through the valve in case it is “blown out” by the aortic stenotic jet. A GooseNeck snare (Medtronic) sized 1:1 to the LVOT diameter is then positioned immediately below the aortic valve, parallel to the aortic annulus plane. To position the aortic catheter (Table 2), two fluoroscopic projections are used: the front view in which the catheter is directed toward the center of the leaflet of interest and the side view in which the catheter is directed toward the base of the leaflet of interest (Figure 1). A 0.014-inch Astato XS 20 guidewire (Asahi Intecc USA, Inc.) is loaded within a PiggyBack wire converter (Teleflex) and advanced through the aortic catheter to the target leaflet hinge-point under angiographic guidance. A scalpel blade is then used to denude the back end of the Astato guidewire, which is then clamped to the electrosurgery pencil set to “pure cut” mode at 30 to 50 W. During cine, the guidewire is electrified for 1 second while being pushed through the PiggyBack and leaflet into the LVOT. The wire is then snared in the LVOT as close to the aortic annulus as possible to avoid mitral chordal entrapment.
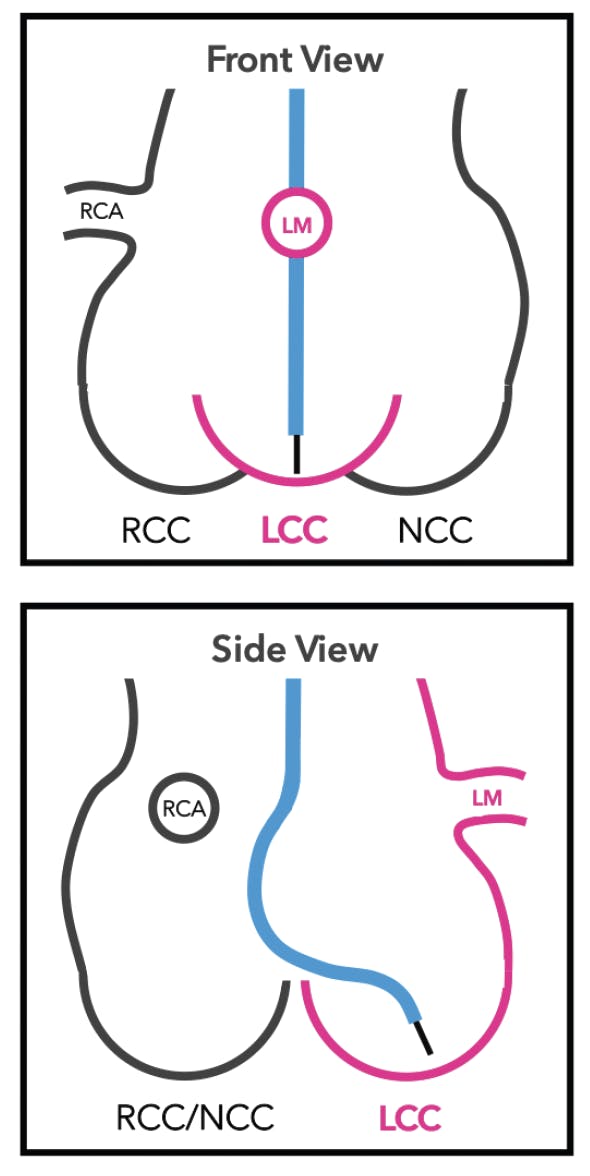
Figure 1. Fluoroscopic projections for BASILICA: an example of cath lab projections (obtained from preprocedure CT) to perform a left leaflet BASILICA. The front view (A) aides in targeting the center of the leaflet, and the side view (B) aides in targeting the base of the leaflet close to the hinge point. LCC, left coronary cusp; LM, left main; NCC, noncoronary cusp; RCA, right coronary artery; RCC, right coronary cusp.
Step 3: Leaflet Laceration
The PiggyBack is then withdrawn outside the body until only 5 to 7 cm of the most proximal Astato guidewire remains exposed. A scalpel blade is then used to denude an approximately 1-mm segment of the guidewire wire just in front of the PiggyBack and on one side (ie, noncircumferential). The denuded segment is then kinked over the back of the scalpel blade. This maneuver creates a “flying V” with the denuded segment on the inner curvature, which will be used for laceration. Then, while pulling the snare from the LVOT catheter, the wire is externalized, and the flying V is advanced into position at the target leaflet. The PiggyBack and Astato are locked into place using double torque devices on both ends. Two large 60-mL syringes filled with 5% dextrose in water are then attached via extension tubing to the sidearms of the aortic and LVOT catheters for injection during leaflet laceration. Dextrose is nonionic, and thus it minimizes leakage current along the length of the guidewire and concentrates charge at the flying V onto the leaflet. Dextrose in water also prevents char and thrombus formation in the aorta during laceration.
At this time, the aortic valve can be recrossed to position a pigtail catheter in the left ventricular apex through the 14- to 18-F DrySeal sheath (alongside the BASILICA catheter). This allows for swift TAVR deployment after BASILICA if necessary. To lacerate, the Astato wire is electrified using “pure cut” mode at 50 to 70 W, and tension is applied from both ends of the guide catheters, while injecting dextrose. Importantly, the operator should not pull too hard as the intention is to cut the leaflet, not to mechanically avulse it. After BASILICA, TAVR is performed as usual. Figure 2 summarizes the key steps of solo BASILICA.

Figure 2. Step-by-step BASILICA procedure on the left aortic leaflet in a patient with a failing 21-mm Mitroflow aortic pericardial heart valve (Sorin Group). An Amplatz Left 2 (AL2) guiding catheter is positioned in the ascending aorta, and a Goose Neck snare alongside a V-18 ControlWire guidewire (Boston Scientific Corporation) is delivered through an MP1 guiding catheter into the LVOT (A). An Astato wire is then advanced through the AL2 toward the base of the left aortic leaflet (B). The Astato wire is then electrified crossing into the LVOT (C). The wire is then snared (D), then the snare is externalized, and the flying V is positioned (E). While injecting 5% dextrose water in both catheters, the Astato wire is electrified to lacerate the leaflet, resulting in leaflet splay (F, G). Aortography demonstrates preserved flow in the left coronary artery after TAVR deployment (H). AVR, aortic valve replacement.
For doppio BASILICA, both catheters for a single leaflet are introduced through the same DrySeal sheath (ie, alongside each other). After positioning the flying V for one leaflet, the entire process is repeated for the second leaflet from the contralateral side. Only once both flying Vs are in place is laceration of one leaflet performed. The first catheter pair is removed, and the aortic valve is recrossed to position a pigtail catheter in the left ventricular apex and allow for swift TAVR deployment after BASILICA. The second laceration is performed after TAVR as usual.
DATA OVERVIEW
BASILICA has been studied comprehensively in a prospective, multicenter, single-arm trial.6,8,9 The BASILICA trial (NCT03381989) is a National Institutes of Health–sponsored investigational device exemption trial examining the safety and feasibility of the BASILICA technique.8,9 Between February and July 2018, the BASILICA trial enrolled 30 patients with severe native or bioprosthetic aortic valve disease who were at high or extreme risk for surgical aortic valve replacement and at high risk of coronary artery obstruction.8,9 BASILICA was successfully performed in 94.7% leaflets (n = 35/37) in 93.3% patients (n = 28/30).8 Leaflet laceration was successful in all leaflets that were successfully traversed.8 All patients had freedom from coronary obstruction, reintervention, or surgery. For patients in whom leaflet traversal was unsuccessful, coronary stents were prepositioned but were only deployed in one patient.8 Leaflet traversal failure was attributed to confluent leaflet calcification at the crossing target at the base of the target leaflet. The primary safety endpoint was met in 70% of patients (n = 21/30), which was predominantly driven by 20% major vascular complications (n = 6/30) that were related to transfemoral TAVR and not BASILICA. At 30 days, myocardial infarction occurred in 3.3% of patients (n = 1/30), stroke occurred in 10% (n = 1/30 disabling stroke; n = 2/30 nondisabling stroke), and death occurred in 3.3% (n = 1/30). Stroke was observed in two of 23 solo BASILICA patients and one of seven doppio BASILICA patients. Stroke was also observed in one of 13 native and two of 17 bioprosthetic BASILICA patients. Cerebral embolic protection was used in 43% of patients. Hypoattenuated leaflet thickening was not identified in any lacerated leaflets 30 days after TAVR. Between 30 days and 1 year, no additional patients had a stroke or myocardial infarction.9 At 1 year, all patients had freedom from aortic valve endocarditis, coronary obstruction, reintervention, or surgery. For those with 1-year echocardiographic assessment (n = 22/27), hemodynamics remained excellent at 1-year follow-up (30-day mean gradient, 15 mm Hg; 1-year mean gradient, 14 mm Hg).
The BASILICA registry is a prospective, multicenter registry of patients at risk for iatrogenic coronary obstruction after TAVR undergoing the BASILICA technique.10 The registry enrolled 214 patients from 25 centers in North America and Europe. Most patients (72.8%) had bioprosthetic aortic valves and underwent solo BASILICA (78.5%). Leaflet traversal and laceration were successful in 94.9% and 94.4% of patients, respectively. Coronary obstruction (partial or complete) was observed in 4.7%. BASILICA procedural success—defined by traversal and laceration without mortality, coronary obstruction, or emergency intervention—was noted in 86.9%. A composite of 30-day mortality and stroke occurred in just 3.4%. Stroke occurred in 2.8%, with just 0.5% disabling stroke at 30 days. Outcomes were comparable between solo and doppio BASILICA, native and bioprosthetic valves, and use and no use of cerebral embolic protection.
FUTURE CHALLENGES
One potential challenge is that the utility of cerebral embolic protection during BASILICA remains uncertain. Additionally, in the BASILICA trial, failure to traverse was attributed to heavy leaflet calcification, and patients with failing transcatheter heart valves were not studied.8
BASILICA may not be effective for redo because of commissural misalignment or limited splaying of lacerated leaflets as they are pinned against the outer frame of the initial transcatheter heart valve.11 However, balloon-assisted BASILICA may be more effective for redo TAVR.12 Dedicated guide catheters13 and electrosurgical guidewires should facilitate wider adoption of BASILICA in the future.
SUMMARY
BASILICA offers a safe, feasible, and efficacious technique to mitigate the risk of iatrogenic coronary obstruction after TAVR. With close adherence to the procedural steps summarized herein, BASILICA can be easily adopted by experienced TAVR operators.
1. Kukucka M, Pasic M, Dreysse S, Hetzer R. Delayed subtotal coronary obstruction after transapical aortic valve implantation. Interact Cardiovasc Thorac Surg. 2011;12:57-60. doi: 10.1510/icvts.2010.252866
2. Ribeiro HB, Nombela-Franco L, Urena M, et al. Coronary obstruction following transcatheter aortic valve implantation: a systematic review. JACC Cardiovasc Interv. 2013;6:452-461. doi: 10.1016/j.jcin.2012.11.014
3. Ribeiro HB, Webb JG, Makkar RR, et al. Predictive factors, management, and clinical outcomes of coronary obstruction following transcatheter aortic valve implantation: insights from a large multicenter registry. J Am Coll Cardiol. 2013;62:1552-1562. doi: 10.1016/j.jacc.2013.07.040
4. Jabbour RJ, Tanaka A, Finkelstein A, et al. Delayed coronary obstruction after transcatheter aortic valve replacement. J Am Coll Cardiol. 2018;71:1513-1524. doi: 10.1016/j.jacc.2018.01.066
5. Ribeiro HB, Rodés-Cabau J, Blanke P, et al. Incidence, predictors, and clinical outcomes of coronary obstruction following transcatheter aortic valve replacement for degenerative bioprosthetic surgical valves: insights from the VIVID registry. Eur Heart J. 2018;39:687-695. doi: 10.1093/eurheartj/ehx455
6. Khan JM, Dvir D, Greenbaum AB, et al. Transcatheter laceration of aortic leaflets to prevent coronary obstruction during transcatheter aortic valve replacement: concept to first-in-human. JACC Cardiovasc Interv. 2018;11:677-689. doi: 10.1016/j.jcin.2018.01.247
7. Lederman RJ, Babaliaros VC, Rogers T, et al. Preventing coronary obstruction during transcatheter aortic valve replacement: from computed tomography to BASILICA. JACC Cardiovasc Interv. 2019;12:1197-1216. doi: 10.1016/j.jcin.2019.04.052
8. Khan JM, Greenbaum AB, Babaliaros VC, et al. The BASILICA trial: prospective multicenter investigation of intentional leaflet laceration to prevent TAVR coronary obstruction. JACC Cardiovasc Interv. 2019;12:1240-1252. doi: 10.1016/j.jcin.2019.03.035
9. Khan JM, Greenbaum AB, Babaliaros VC, et al. BASILICA trial: one-year outcomes of transcatheter electrosurgical leaflet laceration to prevent TAVR coronary obstruction. Circ Cardiovasc Interv. 2021;14:e010238. doi: 10.1161/CIRCINTERVENTIONS.120.010238
10. Khan JM, Babaliaros VC, Greenbaum AB, et al. Preventing coronary obstruction during transcatheter aortic valve replacement: results from the multicenter international BASILICA registry. JACC Cardiovasc Interv. 2021;14:941-948. doi: 10.1016/j.jcin.2021.02.035
11. Khan JM, Bruce CG, Babaliaros VC, et al. TAVR roulette: caution regarding BASILICA laceration for TAVR-in-TAVR. JACC Cardiovasc Interv. 2020;13:787-789. doi: 10.1016/j.jcin.2019.10.010
12. Perdoncin E, Bruce CG, Babaliaros VC, et al. Balloon-augmented leaflet modification with bioprosthetic or native aortic scallop intentional laceration to prevent iatrogenic coronary artery obstruction and laceration of the anterior mitral leaflet to prevent outflow obstruction: benchtop validation and first in-man experience. Circ Cardiovasc Interv. 2021;14:e011028. doi: 10.1161/CIRCINTERVENTIONS.121.011028
13. Lisko JC, Babaliaros VC, Lederman RJ, et al. Pachyderm-shape guiding catheters to simplify BASILICA leaflet traversal. Cardiovasc Revasc Med. 2019;20:782-785.
Advertisement
Advertisement


