Advertisement
Advertisement
October 13, 2023
WhiteSwell’s eLym System Studied in DELTA-HF Early Feasibility Trial
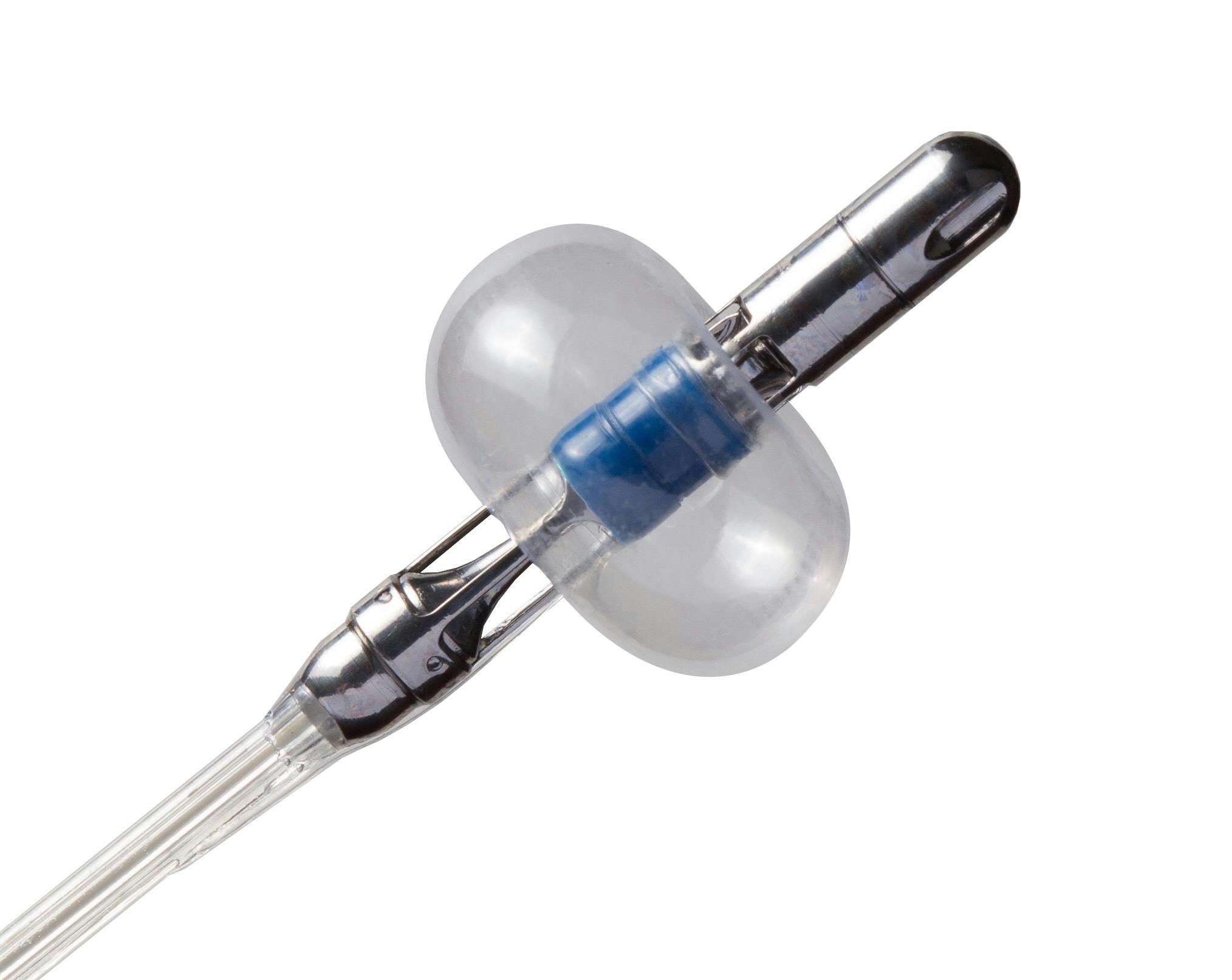
© WhiteSwell, reprinted with permission.
October 13, 2023—WhiteSwell, a privately held company headquartered in Galway, Ireland, and focused on the treatment of acute decompensated heart failure (ADHF), recently announced positive initial results from its ongoing DELTA-HF trial evaluating the WhiteSwell eLym system.
According to the company, the minimally invasive, catheter-based eLym system facilitates the removal of excess fluid from the body’s tissues and organs in patients with ADHF concurrent with diuretic removal of excess vascular fluid.
The eLym system supports the overwhelmed lymphatic system’s ability to actively drain fluid. The device is deployed in the left internal jugular and innominate veins near where the lymphatic system’s major highway, the thoracic duct, connects. It creates a low-pressure zone to facilitate fluid drainage in conjunction with intravenous diuretics.
The DELTA-HF trial is a nonrandomized early feasibility trial evaluating the safety and performance of the eLym system in the treatment of fluid overload or congestion in patients with ADHF. In the trial, nine hospitalized patients received eLym therapy in conjunction with diuretic therapy versus six patients who received standard-of-care treatment with loop diuretics alone.
The DELTA-HF data were presented at HFSA 2023, the annual scientific meeting of the Heart Failure Society of America held October 6-9 in Cleveland, Ohio.
The company reported that treatment with the eLym system was performed safely and successfully with the following findings:
- The device was deployed and activated in nine patients with a mean treatment time of 24 hours
- No patient experienced a serious procedure-, device-, or therapy-related adverse event
Additionally, the company stated that the following early clinical results are promising and support further clinical investigation:
- Patients who underwent therapy with the eLym system plus loop diuretic lost a mean of 6.0 ± 4.6 kg from baseline to hospital discharge while maintaining kidney function, as measured by a stable or improved creatinine (mean Δ -0.10 ± 0.12 mg/dL).
- The loop diuretic-only group lost a mean of 3.3 ± 3.7 kg.
- One of the nine treated patients (11%) was hospitalized within 30 days of discharge.
“It cannot be overemphasized how significant and challenging it is to fully decongest patients hospitalized with decompensated heart failure,” commented William Abraham, MD, in the company’s press release. “Patients with persistent congestion at discharge are more likely to be rehospitalized; are less likely to be treated with optimal medical therapy; and are at greater risk of poor outcomes. Most of the fluid accumulates in the tissues, and we have not previously had a way to actively target that fluid.”
Dr. Abraham, who is Professor of Medicine, Physiology, and Cell Biology at The Ohio State University Wexner Medical Center in Columbus, Ohio, continued, “WhiteSwell’s ingenious approach of reducing thoracic duct pressure to facilitate lymphatic drainage of the tissues enables us to actively address both interstitial tissue and intravascular decongestion simultaneously. I’m particularly encouraged by the amount of decongestion achieved with stable or improved creatinine, an indicator of kidney function.”
Zviad Kereselidze, MD, Head of the Intensive Coronary Care Unit at Tbilisi Heart Center in Tbilisi, Georgia, added, “This is a new approach, and we are excited to investigate WhiteSwell’s catheter-based system. We have treated some of our most challenging patients who are frequently hospitalized with congestion, and we have been pleasantly surprised with how well they have decongested at the time of discharge and how the benefit was sustained at 30-day follow-up.”
WhiteSwell advised that it continues to evaluate this approach to treating ADHF patients with continued enrollment and follow-up in the safety and feasibility DELTA-HF trial of the eLym system.
Advertisement
Advertisement
