Advertisement
Advertisement
April 28, 2025
Solo Pace’s Temporary Pacing System for TAVR Procedures Cleared by FDA
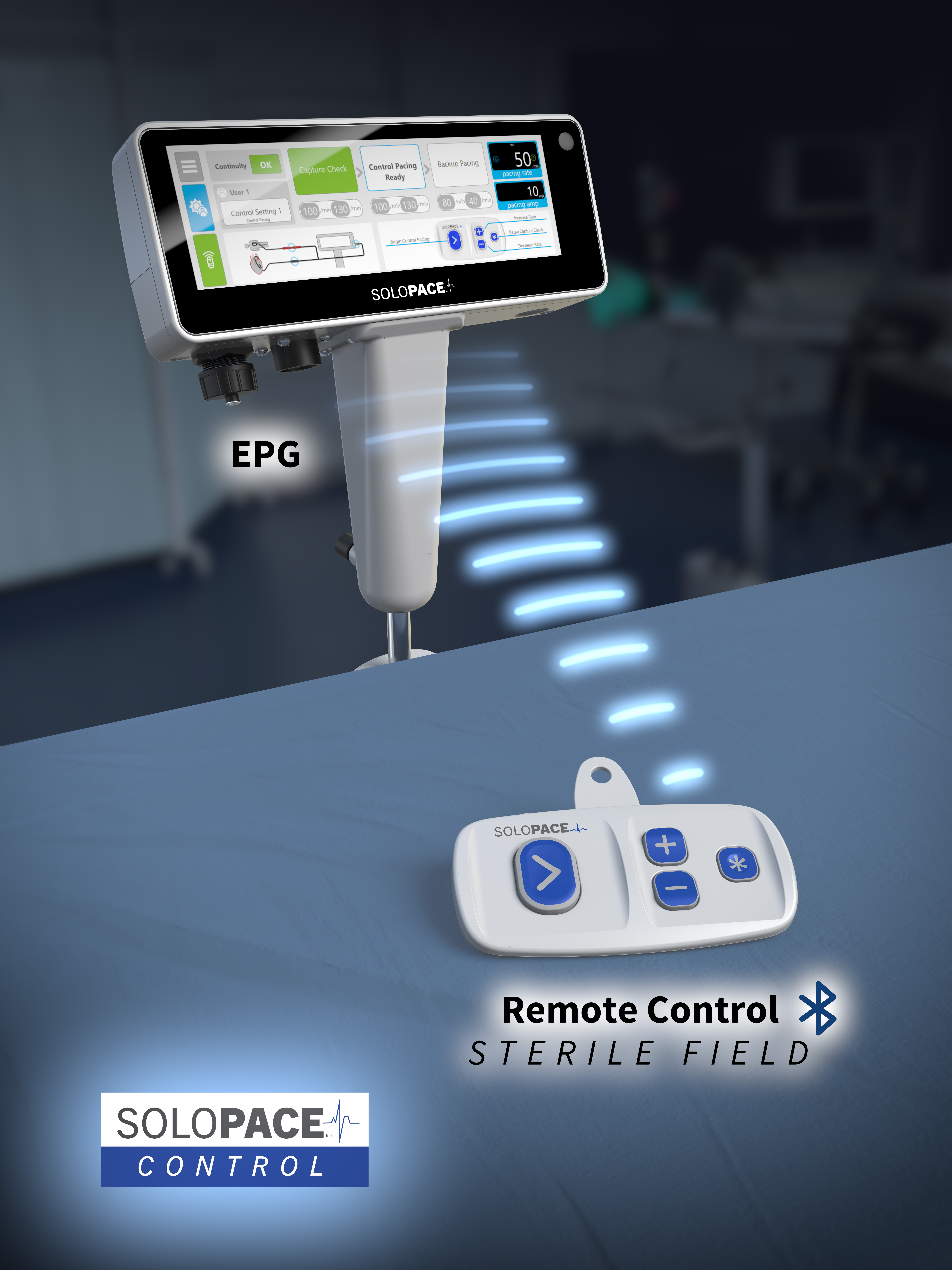 April 28, 2025—Solo Pace Incorporated recently announced FDA clearance and the first-in-human use of the company’s SoloPace control system for temporary pacing in transcatheter aortic valve replacement (TAVR) procedures.
April 28, 2025—Solo Pace Incorporated recently announced FDA clearance and the first-in-human use of the company’s SoloPace control system for temporary pacing in transcatheter aortic valve replacement (TAVR) procedures.
According to the company, the SoloPace control system is engineered to improve TAVR temporary pacing efficiency and reduce patient risks. The device features operator-specific protocols, automated pace capture checks, an intuitive interface, and sterile remote-control operation. In addition, the system is compatible with patient cables for all common procedural pacing methods, noted the company.
Solo Pace stated that the initial cases using the device were completed by Chief of Cardiology Paul Teirstein, MD, and interventional cardiologist Curtiss Stinis, MD, at Scripps Clinic in La Jolla, California.
“Solo Pace has modernized pacing during TAVR valve deployment,” commented Dr. Teirstein in the company’s press release. “The device takes variability out of the procedure by giving the operator full control while automating ramp-up and back-up algorithms.”
Dr. Teirstein continued, “The TAVR procedure is simplified, reducing physician and staff workload. We preprogram pacer settings to reliably deliver the same procedure every time. This allows us to focus on the patient and free up resources, especially at centers where nurses and technologists currently control the pacemaker generator.”
Advertisement
Advertisement
