Advertisement
Advertisement
April 18, 2024
RCE’s Infrasensor Wearable Device Studied for Continuous Cardiac Biomarker Monitoring
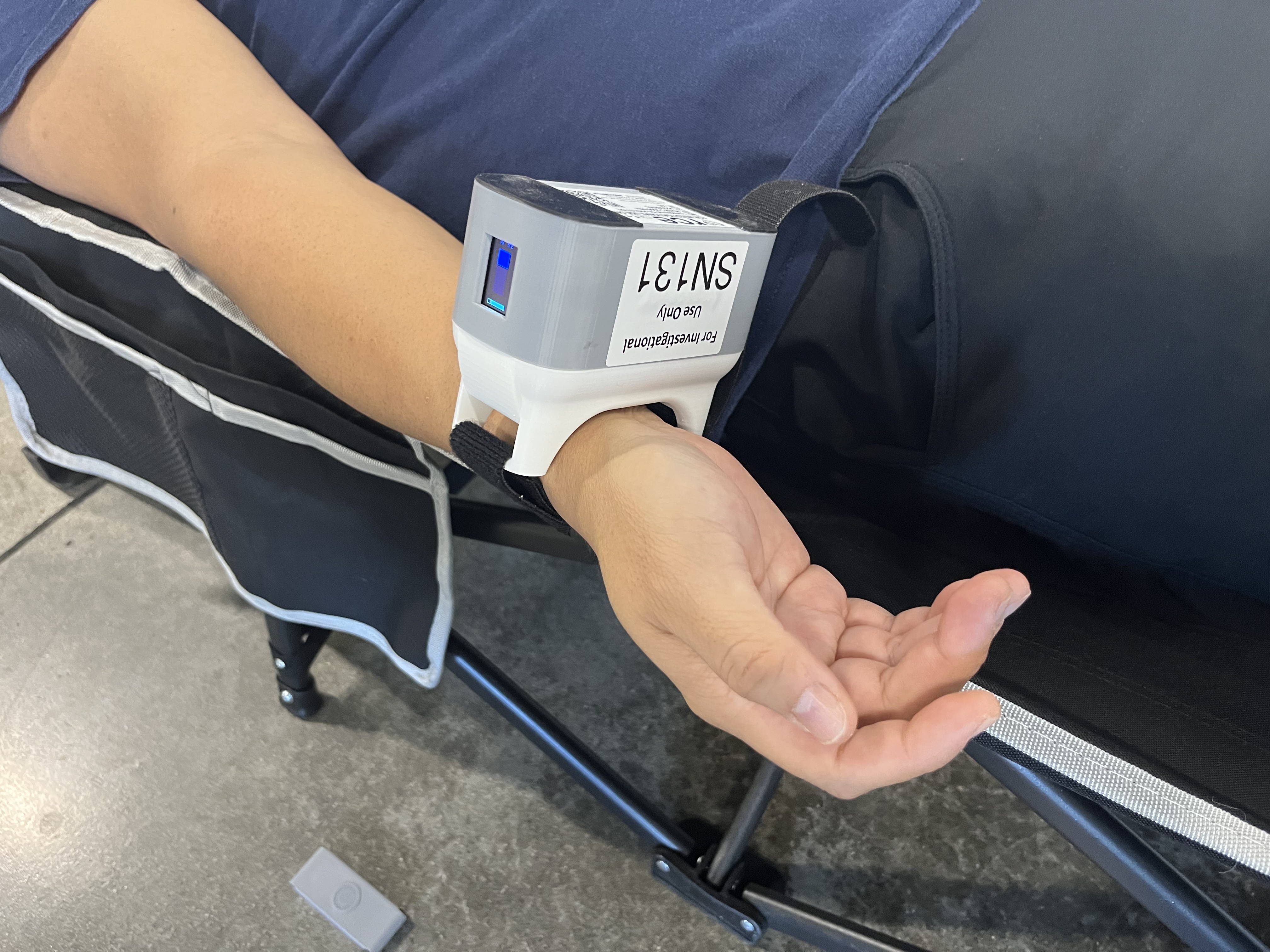 April 18, 2024—RCE Technologies, Inc. announced results were presented from a pilot study enrolling cardiac catheterization patients in a first-in-human trial of transdermal continuous cardiac biomarker monitoring using the company’s Infrasensor wearable device. RCE is a California-based medical technology company using artificial intelligence–based technology for early heart attack detection.
April 18, 2024—RCE Technologies, Inc. announced results were presented from a pilot study enrolling cardiac catheterization patients in a first-in-human trial of transdermal continuous cardiac biomarker monitoring using the company’s Infrasensor wearable device. RCE is a California-based medical technology company using artificial intelligence–based technology for early heart attack detection.
According to RCE, investigators found that the Infrasensor device enabled real-time continuous monitoring throughout the coronary catheterization, extended monitoring in the postanesthesia care unit, and removal just before hospital discharge. The study was composed of 20 consecutive patients, both those scheduled diagnostic catheterization procedures and emergency cases of heart attacks. Clinical staff applied the Infrasensor wearable device to patients’ wrists before the procedure.
The study was conducted by Josiah N. Brown, MD; Ronald P. Karlsberg, MD; and Suhail Y. Dohad, MD, with Cedars Sinai Medical Center in Los Angeles, California.
The study was featured in a “Spotlight on Special Topics” presentation (#1542-148) of advancements in cardiac monitoring technology at ACC.24, the American College of Cardiology (ACC)’s annual scientific session held April 6-8 in Atlanta, Georgia. The study abstract is available online in a meeting supplement of the Journal of the American College of Cardiology (JACC; 2024;83:2685).
“This is an innovative platform for measuring biomarkers,” commented Dr. Karlsberg in the company’s press release. “The insights underscore the significant promise of this novel technology, which inaugurates a new era for monitoring in a wide variety of scenarios real-time and affords the opportunity to develop and refine diagnostic pathways that will have a substantial impact on patient care.”
As summarized in the JACC abstract, of the 20 patients undergoing percutaneous coronary interventions in the study, five had stable coronary artery disease and 15 were experiencing acute coronary syndrome. The RCE transdermal wearable sensor gathered data every 3 minutes during the procedure until discharge or 4 hours postprocedure. Readings were compared with serum troponin-l levels at beginning and end. Sensor data was then correlated with procedural details and troponin-l levels.
RCE noted that the study builds on earlier findings that were announced announced by ACC in March 2023. That study posited that exploring real-time biomarker trends from the wrist wearable could further the understanding of the transdermal signal and the association with clinically relevant use cases at the point of care. Partho P. Sengupta, MD, presented the study at ACC.23/WCC, the ACC’s annual scientific session together with the World Congress of Cardiology held March 4-6, 2023, in New Orleans, Louisiana. The study was simultaneously published by Shantanu Sengupta, MD, et al in the European Heart Journal - Digital Health (2023;4:245-154).
Advertisement
Advertisement
