Advertisement
Advertisement
May 11, 2026
Enrollment Completed in FIH Study of Robocath’s Next-Generation Robotic Platform for PCI
KEY TAKEAWAYS
- Robocath completes enrollment in FIH clinical study of next-generation system for robot-assisted PCI.
- The 20-patient study is being conducted by Mohammed Nejjari, MD, at the Centre Cardiologique du Nord in Saint-Denis, France
- Promising technical outcomes observed across all lesion subgroups:type A/B1, type B2, and type C.
May 11, 2026—Robocath, which is based in Rouen, France, announced the completion of patient enrollment in the first-in-human (FIH) clinical study evaluating the feasibility and safety of its next-generation robotic system for robot-assisted percutaneous coronary intervention (RA-PCI) in patients with coronary artery disease.
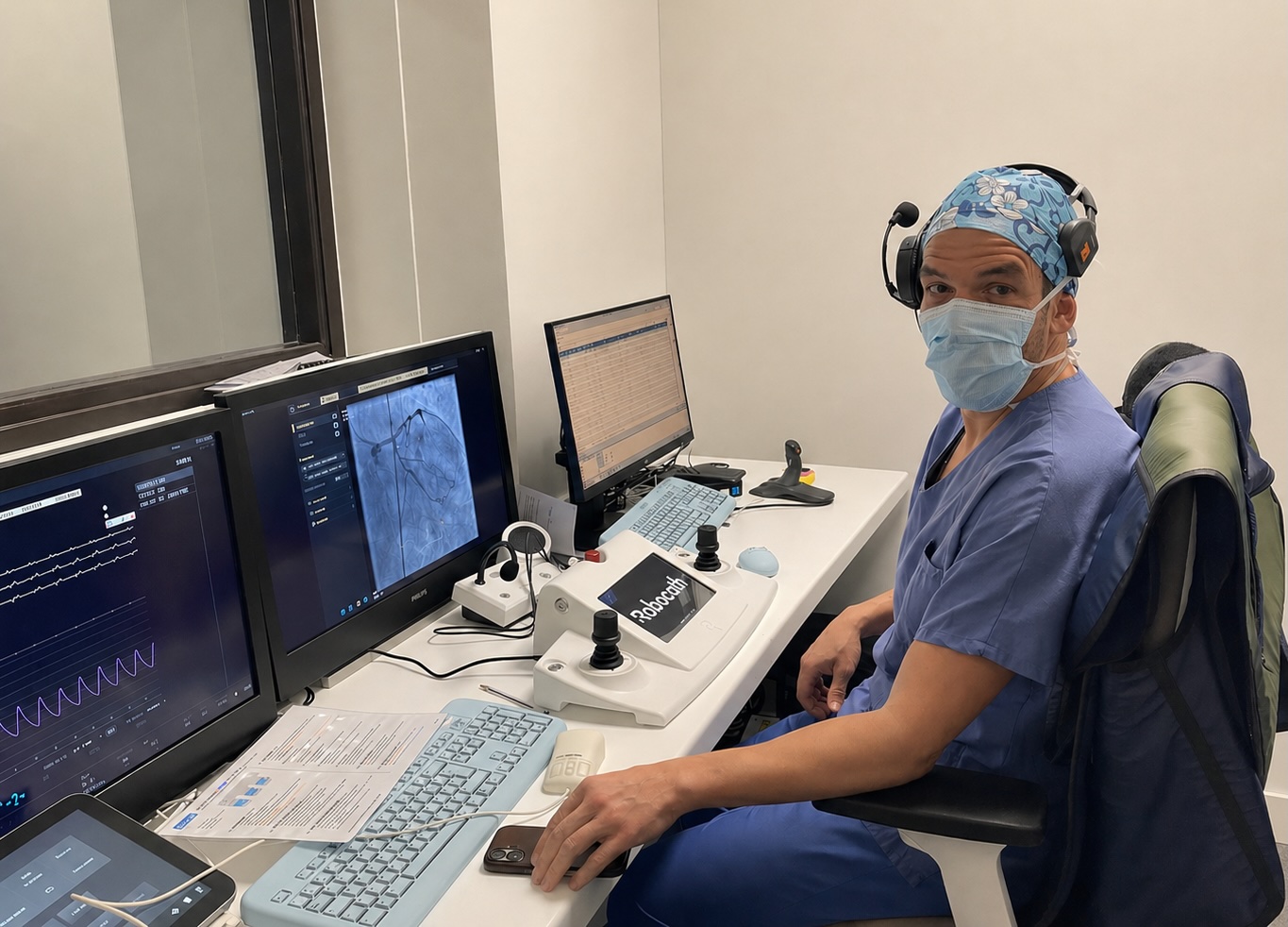
Principal Investigator Mohammed Nejjari, MD
As noted in the press release, the study included 20 patients (age, ≥ 18 years) with coronary artery disease, primarily complex cases seen in real-world catheterization laboratory practice. The patients were stratified by coronary lesion complexity across three subgroups: type A/B1 lesions (6 patients); type B2 lesions (7 patients;) and type C lesions (7 patients). The commencement of the study was announced in January 2026.
The company advised that to date, no safety concerns have been reported in connection with the use of the Robocath system for RA-PCI. Additionally, technical outcomes observed across all lesion subgroups—including complex type B2 and type C anatomies—are considered promising.
The study was conducted at the Centre Cardiologique du Nord in Saint-Denis, France. It was led by Principal Investigator Mohammed Nejjari, MD, and coinvestigator Franck Digne, MD.
“Completing enrollment in this FIH study is a defining moment in the field of interventional cardiology,” commented Dr. Nejjari in Robocath’s press release. “For the first time, we have demonstrated that robotic assistance can be safely and reliably applied to the full spectrum of coronary lesion complexity, including the most challenging anatomies. This is not a glimpse into the future: it is the future, starting now.”
Full data analysis and detailed results will be presented in the near-term in forthcoming scientific communications, stated Robocath.
In April 2026, a definitive agreement to acquire Robocath was announced by Stereotaxis, a St. Louis, Missouri–based company focused on robotics for endovascular intervention.
Advertisement
Advertisement
