Advertisement
Advertisement
December 5, 2022
Abiomed’s Impella RP Flex With SmartAssist Used in First Postapproval Procedures
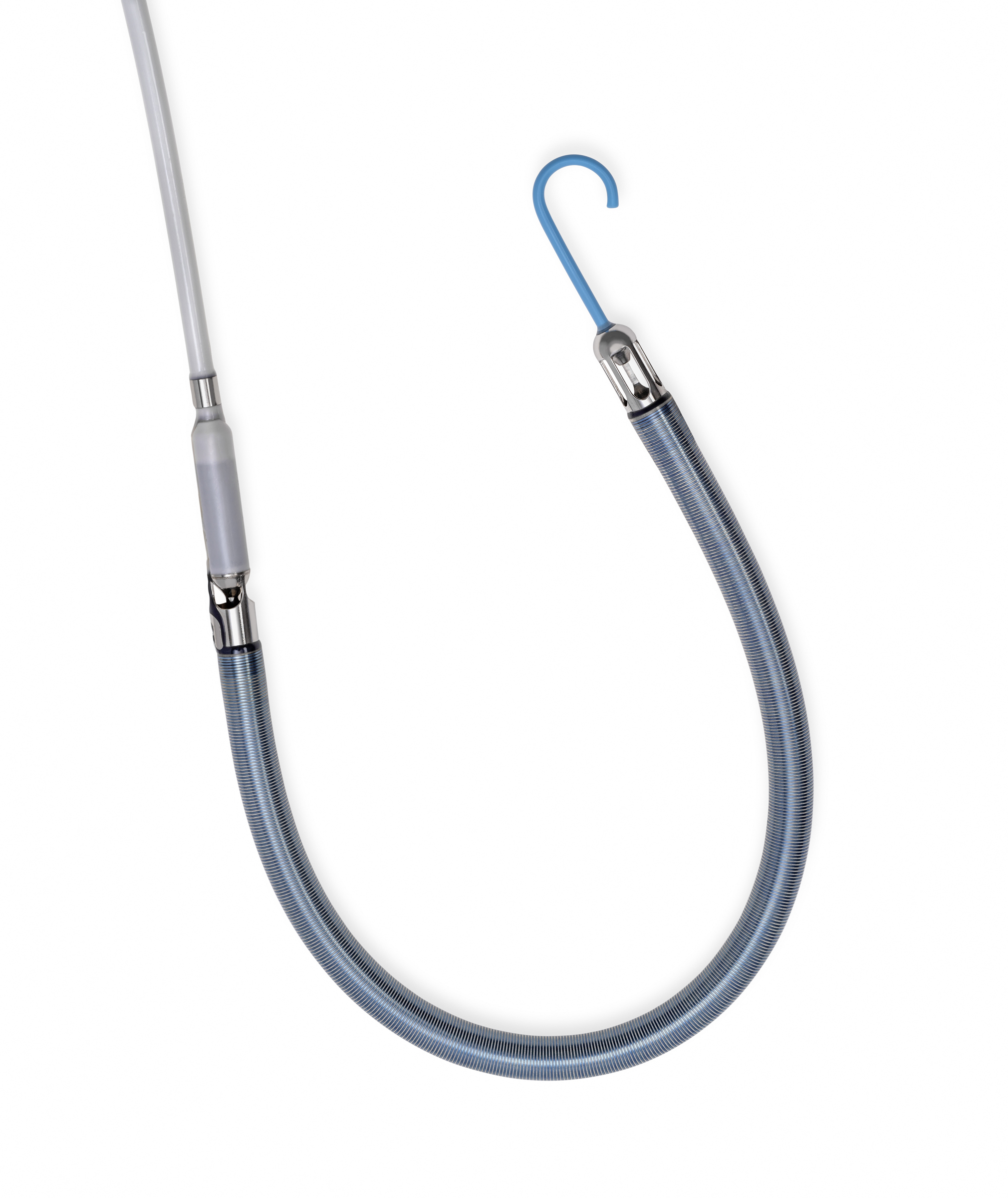 December 5, 2022—Abiomed announced the first three patients have been treated with Impella RP Flex with SmartAssist, the company’s newest heart pump for patients experiencing right heart failure. All three patients have been successfully weaned off Impella support and two have returned home with their native hearts. The patients were treated at Hackensack University Medical Center/Hackensack Meridian Health in Hackensack, New Jersey, and Kingwood Medical Center in Kingwood, Texas.
December 5, 2022—Abiomed announced the first three patients have been treated with Impella RP Flex with SmartAssist, the company’s newest heart pump for patients experiencing right heart failure. All three patients have been successfully weaned off Impella support and two have returned home with their native hearts. The patients were treated at Hackensack University Medical Center/Hackensack Meridian Health in Hackensack, New Jersey, and Kingwood Medical Center in Kingwood, Texas.
According to the company, Impella RP Flex is the latest iteration of the Impella RP heart pump, which received FDA approval under a humanitarian device exemption (HDE) in 2015, which was followed by premarket approval in 2017. Impella RP with SmartAssist is FDA approved to treat right heart failure or decompensation following left ventricular assist device implantation, myocardial infarction, heart transplant, or open-heart surgery.
In October 2022, Abiomed announced that Impella RP Flex with SmartAssist received FDA approval to provide temporary right ventricular support for up to 14 days in patients with a body surface area ≥ 1.5 m2, who develop acute right heart failure or decompensation following left ventricular assist device implantation, myocardial infarction, heart transplant, or open-heart surgery.
Impella RP Flex is being introduced in the United States through a controlled rollout at leading centers for heart recovery.
Mark Anderson, MD, chairman of the department of cardiac surgery and a cardiothoracic surgeon at the Heart and Vascular Hospital at Hackensack University Medical Center/Hackensack Meridian Health, led the medical team for the world’s first Impella RP Flex implant.
Dr. Anderson and his colleague Yuriy Dudiy, MD, implanted Impella RP Flex in a patient (age, 71 years), who was having a minimally invasive valve surgery. The patient received Impella RP Flex support for 5 days while the heart rested and recovered. Dr. Anderson’s second Impella RP Flex case involved a patient (age, 51 years), who experienced cardiac arrest and received Impella support for 4 days after valve surgery.
“Impella RP Flex is an innovative technology that can allow right heart failure patients to be mobile while on support,” commented Dr. Anderson in Abiomed’s press release.
Robert Salazar, MD, an interventional cardiologist and Director of Cardiovascular Research at Kingwood Medical Center, with his colleague Marloe Prince, MD, implanted Impella RP Flex in a patient (age, 75 years) after a thrombectomy procedure for a pulmonary embolism. The patient remained on support for 4 days and is expected to be discharged from the hospital in the coming days.
Dr. Salazar commented in the company’s press release, “Right ventricular failure is important to identify and treat early, and Impella RP Flex will enable more patients to achieve native heart recovery.”
According to Abiomed, Impella RP Flex is implanted percutaneously through the internal jugular vein, which provides the option for patient mobility while on support, and it is designed to be easy to implant, with a flexible canula that is advanced over an extrasupportive guidewire. It includes the company’s SmartAssist dual-sensor technology with Impella Connect, giving health care providers the ability to monitor the pump remotely from any internet-connected device and providing advanced metrics to help with pump management and weaning. It is also compatible with a sodium bicarbonate purge solution to simplify patient anticoagulation management.
In October 2022, the company announced that the FDA accepted and closed the Impella RP postapproval study, which enrolled 110 patients at 29 study sites. The study represented real-world experience and identified the best practice of treating right heart failure early.
The company advised that in 2018, data showed that patients who received Impella RP support within 48 hours of cardiogenic shock onset had a significantly higher survival rate than those who received delayed right-heart support (72% vs 14%; P < .001). The findings were published by Dr. Anderson et al in the Journal of Heart and Lung Transplantation (2018;37:1448-1458).
Advertisement
Advertisement
